Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
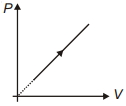
- 1Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
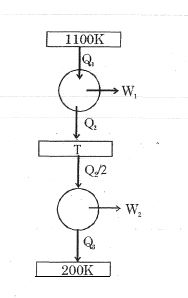
- 2Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
- 3View SolutionA measure of the degree of disorder of a system is known as
- 4The temperature of sink of Carnot engine is ${27^o}C$. Efficiency of engine is $25\%.$ Then temperature of source is ...... $^oC$View Solution
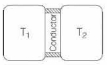
- 5A thin piece of thermal conductor of constant thermal conductivity insulated on the lateral sides connects two reservoirs which are maintained at temperatures $T_{1}$ and $T_{2}$ as shown in the figure alongside. Assuming that the system is in steady state, which of the following plots best represents the dependence of the rate of change of entropy on the ratio of $T_{1} / T_{2}$ ?View Solution
- 6A closed vessel contains $0.1$ mole of a monoatomic ideal gas at $200\, K$. If $0.05$ mole of the same gas at $400\, K$ is added to it, the final equilibrium temperature (in $K$ ) of the gas in the vessel will be closed toView Solution
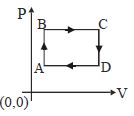
- 7The figure shows $P-V$ diagram of a thermodynamic cycle. Which corresponding curve is correct?View Solution
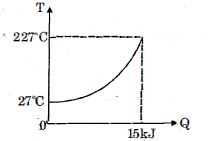
- 8$Q$ amount of heat is given to $0.5\ mole$ of an ide al mono-atomic gas by a process $TV^n$ constant. Following graph shows variation of temperature with $Q$ . Find value of $n$.View Solution
- 9A total of $48 \mathrm{~J}$ heat is given to one mole of helium kept in a cylinder. The temperature of helium increases by $2^{\circ} \mathrm{C}$. The work done by the gas is : (Given, $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$.)View Solution
- 10View SolutionDuring the thermodynamic process shown in figure for an ideal gas