An ideal gas $(\gamma = 1.5)$ is expanded adiabatically. How many times has the gas to be expanded to reduce the root mean square velocity of molecules $2.0$ times
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Nitrogen gas $N _2$ of mass $28 \,g$ is kept in a vessel at pressure of $10 \,atm$ and temperature $57^{\circ} C$. Due to leakage of $N _2$ gas its pressure falls to $5 \,atm$ and temperature to $27^{\circ} C$. The amount of $N _2$ gas leaked out is .......... $g$View Solution
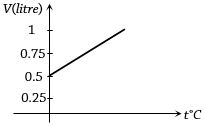
- 2Graph between volume and temperature for a gas is shown in figure. If $\alpha$ = Volume coefficient of gas = $\frac{1}{273}$ per $^o C$,then what is the volume of gas at a temperature of $819 ^o C$View Solution
- 3If the $rms$ speed of oxygen molecules at $0^{\circ} {C}$ is $160\; {m} / {s}$, find the rms speed of hydrogen molecules at $0^{\circ} {C}$. (In ${m}/{s}$)View Solution
- 4Under an adiabatic process, the volume of an ideal gas gets doubled. Consequently the mean collision time between the gas molecule changes from $\tau_{1}$ to $\tau_{2} .$ If $\frac{C_{p}}{C_{v}}=\gamma$ for this gas then a good estimate for $\frac{\tau_{2}}{\tau_{1}}$ is given by :View Solution
- 5The temperature at which the root mean square velocity of a molecule will be doubled than at $100°C$View Solution
- 6The specific heat at constant volume for the monoatomic argon is $0.075\, kcal/kg-K,$ whereas its gram molecular specific heat ${C_V}$ $= 2.98\, cal/mole/K.$ The mass of the argon atom isView Solution
$(Avogadro’s \,number = 6.02 \times {10^{23}}\,molecules/mole)$
- 7A hot air balloon with a payload rises in the air. Assume that the balloon is spherical in shape with diameter of $11.7 \,m$ and the mass of the balloon and the payload (without the hot air inside) is $210 \,kg$. Temperature and pressure of outside air are $27^{\circ} C$ and $1 atm =10^5 \,N / m ^2$, respectively. Molar mass of dry air is $30 \,g$. The temperature of the hot air inside is close to .......... $^{\circ} C$ [The gas constant, $R=8.31 \,JK ^{-1} mol ^{-1}$ ]View Solution
- 8A gas mixture consists of $2$ moles of oxygen and 4 moles of neon at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system will be $...........\,RT$View Solution
- 9The ratio of mean kinetic energy of hydrogen and nitrogen at temperature $300 \,K$ and $450\, K$ respectively isView Solution
- 10The root mean square velocity of a gas molecule of mass $m$ at a given temperature is proportional toView Solution
