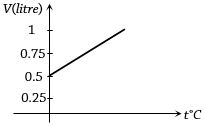
Graph between volume and temperature for a gas is shown in figure. If $\alpha$ = Volume coefficient of gas = $\frac{1}{273}$ per $^o C$,then what is the volume of gas at a temperature of $819 ^o C$

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If $\alpha$ moles of a monoatomic gas are mixed with $\beta$ moles of a polyatomic gas and mixture behaves like diatomic gas, then [neglect the vibrational mode of freedom]View Solution
- 2A mixture of hydrogen and oxygen has volume $2000 \; cm ^{3}$, temperature $300 \; K$, pressure $100 \; kPa$ and mass $0.76 \; g$ The ratio of number of moles of hydrogen to number of moles of oxygen in the mixture will beView Solution
- 3On $0^\circ C$ pressure measured by barometer is $760\, mm.$ What will be pressure on $100^\circ C$View Solution
- 4View SolutionWhich of the following graphs represent the behavior of an ideal gas ? Symbols have their usual meaning.
- 5At which of the following temperature would the molecules of a gas have twice the average kinetic energy they have at $20°C$View Solution
- 6The r.m.s. velocity of a gas at a certain temperature is $\sqrt 2 $ times than that of the oxygen molecules at that temperature. The gas can beView Solution
- 7The respective speeds of the molecules are $1, 2, 3, 4$ and $5\, km/sec.$ The ratio of their $r.m.s. $ velocity and the average velocity will beView Solution
- 8A diatomic gas molecule has translational, rotational and vibrational degrees of freedom. The ${C_P}/{C_V}$ isView Solution
- 9A vessel of volume $0.02 \,\,m^3$ contains a mixture of hydrogen and helium at $20\,^oC$ and $2$ atmospheric pressure. The mass of mixture is $5\,\, gms$. Find the ratio of mass of hydrogen to that of helium in the mixture.View Solution
- 10Molecules of an ideal gas are known to have three translational degrees of freedom and two rotational degrees of freedom. The gas is maintained at a temperature of $T$. The total internal energy, $U$ of a mole of this gas, and the value of $\gamma\left(=\frac{ C _{ P }}{ C _{ v }}\right)$ given, respectively, byView Solution
