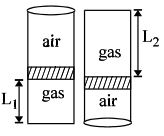
An ideal gas is trapped inside a test tube of cross-sectional area $20 \times 10^{-6} \,\,m^2$ as shown in the figure. The gas occupies a height $L_1$ at the bottom of the tube and is separated from air at atmospheric pressure by a mercury column of mass $0.002\,\, kg$. If the tube is quickly turned isothermally, upside down so that $L_2$ mercury column encloses the gas from below. The gas now occupies height $L_1$ in the tube. The ratio $L_1$ is [Take atmospheric pressure $= 10^5 Nm^{-2}]$

Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The ratio of mean kinetic energy of hydrogen and nitrogen at temperature $300 \,K$ and $450\, K$ respectively isView Solution
- 2The graph between pressure $(P)$ and $\frac{1}{V}$ ($V$ is volume) for constant temperature process in ideal equation $(PV = nRT)$View Solution
- 3Two identical bodies are made of a material for which the heat capacity increases with temperature. One of these is held at a temperature of $100^{\circ} C$, while the other one is kept at $0^{\circ} C$. If the two are brought into contact, then assuming no heat loss to the environment, the final temperature that they will reach isView Solution
- 4View SolutionOn absolute temperature, the kinetic energy of the molecules
- 5The temperature of an ideal gas is increased from $200\,K$ to $800\,K$. If r.m.s. speed of gas at $200\,K$ is $v_0$. Then, r.m.s. speed of the gas at $800\,K$ will be:View Solution
- 6A gas obeying the equation of state $p V=R T$ undergoes a hypothetical reversible process described by the equation, $p V^{5 / 3} \exp \left(-\frac{p V}{E_{0}}\right)=C_{1}$, where $C_{1}$ and $E_{0}$ are dimensioned constants. Then, for this process, the thermal compressibility at high temperatureView Solution
- 7When an air bubble of radius $‘r’$ rises from the bottom to the surface of a lake, its radius becomes $5r/4$ (the pressure of the atmosphere is equal to the $10 \,m$ height of water column). If the temperature is constant and the surface tension is neglected, the depth of the lake is .... $m$View Solution
- 8View SolutionAt a given volume and temperature, the pressure of a gas
- 9View SolutionThe product of the pressure and volume of an ideal gas is
- 10If $\alpha$ moles of a monoatomic gas are mixed with $\beta$ moles of a polyatomic gas and mixture behaves like diatomic gas, then [neglect the vibrational mode of freedom]View Solution
