The product of the pressure and volume of an ideal gas is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA container with rigid walls is covered with perfectly insulating material. The container is divided into two parts by a partition. One part contains a gas while the other is fully evacuated (vacuum). The partition is suddenly removed. The gas rushes to fill the entire volume and comes to equilibrium after a little time. If the gas is not ideal, then
- 2On giving equal amount of heat at constant volume to $1\, mol$ of a monoatomic and a diatomic gas the rise in temperature $(\Delta T)$ is more forView Solution
- 3View SolutionWhat will be the effect on the root mean square velocity of oxygen molecules if the temperature is doubled and oxygen molecule dissociates into atomic oxygen?
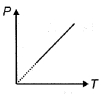
- 4The pressure $P$ of an ideal diatomic gas varies with its absolute temperature $T$ as shown in figure. The molar heat capacity of gas during this process is ........... $R$ [$R$ is gas constant]View Solution
- 5One kg of a diatomic gas is at a pressure of $8 × 10^4\ N/m^2$. The density of the gas is $4\ kg/m^3$. What is the energy of the gas due to its thermal motion ?View Solution
- 6At temperature $T,$ the $r.m.s.$ speed of helium molecules is the same as $r.m.s.$ speed of hydrogen molecules at normal temperature and pressure. The value of $T$ is ....... $^oC$View Solution
- 7$3\,moles$ of an ideal gas at a temperature of $27^{\circ}\,C$ are mixed with $2\,moles$ of an ideal gas at a temperature $227^{\circ}\,C$, determine the equilibrium temperature (${}^o C$) of the mixture, assuming no loss of energy.View Solution
- 8The mean kinetic energy of a gas at $300\, K$ is $100\, J.$ The mean energy of the gas at $450\, K$ is equal to ...... $J$View Solution
- 9The volume occupied by the molecules contained in $4.5\,kg$ water at $STP$, if the intermolecular forces vanish away is ........ $m ^{3}$View Solution
- 10View SolutionThe quantity of heat required to raise one mole through one degree Kelvin for a monoatomic gas at constant volume is
