On absolute temperature, the kinetic energy of the molecules
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe molecules of air in the room that you are sitting are all experiencing the force of gravity tending to bring them down. The molecules are also frequently and randomly undergoing collisions, which tend to oppose the effect of fall under gravity. The density of air is nearly uniform throughout the room because
- 2View SolutionIf a Vander-Waal's gas expands freely, then final temperature is
- 3At constant volume the specific heat of a gas is $\frac{{3R}}{2}$, then the value of $'\gamma '$ will be ....View Solution
- 4View SolutionThe kinetic energy per gm mol for a diatomic gas at room temperature is
- 5A box containing $N$ molecules of a perfect gas at temperature ${T_1}$ and pressure ${P_1}$. The number of molecules in the box is doubled keeping the total kinetic energy of the gas same as before. If the new pressure is ${P_2}$ and temperature ${T_2}$, thenView Solution
- 6View SolutionThe number of translational degrees of freedom for a diatomic gas is
- 7The total kinetic energy of $1$ mole of oxygen at $27^{\circ} \mathrm{C}$ is :View Solution
[Use universal gas constant $(R)=8.31 \mathrm{~J} / \mathrm{mole} \mathrm{K}$ ]
- 8A volume $V$ and pressure $P$ diagram was obtained from state $1$ to state $2$ when a given mass of a gas is subjected to temperature changes. During this process the gas isView Solution
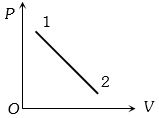
- 9A certain amount of an ideal monatomic gas needs $20 \,J$ of heat energy to raise its temperature by $10^{\circ} C$ at constant pressure. The heat needed for the same temperature rise at constant volume will be ........ $J$View Solution
- 10A sealed container with negligible coefficient of volumetric expansion contains helium (a monoatomic gas). When it is heated from $300\, K$ to $600\, K,$ the average $K.E.$ of helium atoms isView Solution
