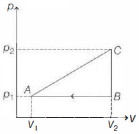
As $B \rightarrow A \rightarrow C$ is a closed cyclic process, we have
$\Delta U($ complete cycle $)=0$
So, by first law of thermodynamics,
we have $\Delta Q=\Delta W$
or $\Delta Q_{A B}+\Delta Q_{B C}+\Delta Q_{A C}=\Delta W$
$=\Delta W_{A B}+\Delta W_{B C}+\Delta W_{A C}$
$=$ Area enclosed under $p-V$ graph
$\Rightarrow \Delta Q_{A C}=-\left(\Delta Q_{A B}\right.\left.+\Delta Q_{B C}\right)+\frac{1}{2}$ $\left(V_2-V_1\right)\left(p_2-p_1\right)$ $=-\left(\frac{3}{2} n R \Delta T-p_1\left(V_2-V_1\right)\right.$ $\left.+\frac{3}{2} n R \Delta T-V_2\left(p_2-p_1\right)\right)$ $+\frac{1}{2}\left(V_2-V_1\right)\left(p_2-p_1\right)$
$\therefore \Delta Q_{A C}=2\left(p_2 V_2-\right.\left.p_1 V_1\right)$ $+\frac{1}{2}\left(V_2-V_1\right)\left(p_2-p_1\right)$
Download our appand get started for free
Similar Questions
- 1If one mole of an ideal gas goes through the process $A \rightarrow B$ and $B \rightarrow C .$ Given that $T _{ A }=400\, K ,$ and $T _{ C }=400 \,K .$ If $\frac{ P _{ B }}{ P _{ A }}=\frac{1}{5},$ then find the heat supplied to the gas (in $J$)View Solution
- 2An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution
- 3A polyatomic gas $\left( {\gamma = \frac{4}{3}} \right)$ is compressed to $\frac{1}{8}$ of its volume adiabatically. If its initial pressure is ${P_o}$, its new pressure will beView Solution
- 4For an ideal gas, the internal energy is given by $U=5 p V / 2+C$, where $C$ is a constant. The equation of the adiabats in the $p V$-plane will beView Solution
- 5During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of $\frac{{{C_P}}}{{{C_V}}}$ for the gas isView Solution
- 6A monoatomic gas is taken along path $AB$ as shown. Calculate change in internal energy of systemView Solution
- 7A sample of gas at temperature $\mathrm{T}$ is adiabatically expanded to double its volume. Adiabatic constant for the gas is $\gamma=3 / 2$. The work done by the gas in the process is : $(\mu=1 \mathrm{~mole})$View Solution
- 8An ideal gas expands according to the law $P^2 V=$ constant. The internal energy of the gasView Solution
- 9The volume of a gas is reduced adiabatically to $\frac{1}{4}$ of its volume at $27°C$, if the value of $\gamma = 1.4,$ then the new temperature will beView Solution
- 10A Carnot's engine working between $400\, K$ and $800\, K$ has a work output of $1200\, J$ per cycle. The amount of heat energy supplied to the engine from the source in each cycle is ........... $J$View Solution
