An ideal gas expands according to the law $P^2 V=$ constant. The internal energy of the gas
Diffcult
(a)
$P^2 V=K$
or $P V^{-2}=K$
$N=-2$
$C=C_V+\frac{R}{1-N}=$ positive quantity
$C > 0$
$W > 0 \quad$ [gas is expanding]
$\because P V^{-2}=K$ so $T V^{-3}=$ constant
$\Rightarrow T$ will increases if $V$ increases.
$\Rightarrow \Delta T > 0$
So $\Delta U=\eta C \Delta T > 0$
It will increase continuously.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ Air quickly leaking out of a balloon becomes cooler.View Solution
$Reason :$ The leaking air undergoes adiabatic expansion. - 2During the melting of a slab of ice at $273\, K$ at atmospheric pressureView Solution
- 3$N _{2}$ gas is heated from $300\, K$ temperature to $600\, K$ through an isobaric process. Then find the change in entropy of the gas. $( n =1 mole )$ (in $J/K$)View Solution
- 4In a thermodynamic process, pressure of a fixed mass of a gas is changed in such a manner that the gas molecules gives out $20 J$ of heat and $10 J$ of work is done on the gas. If the initial internal energy of the gas was $40 J,$ then the final internal energy will be ........ $J$View Solution
- 5View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
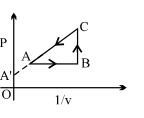
- 6When a system is taken from state $i$ to a state $f$ along path $iaf, \,Q = 50\,J$ and $W = 20J.$ Along path $ibf, \,Q = 35J.$ If $W = - 13J$ for the curved return path $f i, Q$ for this path is ...... $J$View Solution
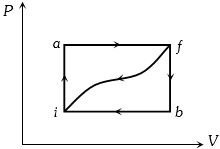
- 7An ideal heat engine exhausting heat at $77\,^oC$. To have a $30\%$ efficiency. It must take heat at...... $^oC$View Solution
- 8Carnot cycle is plotted in $P-V$ graph. Which portion represents an isothermal expansion?View Solution
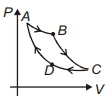
- 9An ideal gas is taken from state $1$ to state $2$ through optional path $A, B, C \& D$ as shown in $P-V$ diagram. Let $Q, W$ and $U$ represent the heat supplied, work done $\&$ internal energy of the gas respectively. ThenView Solution
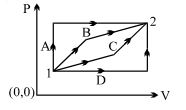
- 10A given ideal gas with $\gamma = \frac{{{C_p}}}{{{C_v}}} = 1.5$ at a temperature $T$. If the gas is compressed adiabatically to one-fourth of its initial volume, the final temperature will be ..... $T$View Solution
