An ideal heat engine exhausting heat at $77\,^oC$. To have a $30\%$ efficiency. It must take heat at...... $^oC$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
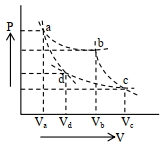
- 1Two different adiabatic paths for the same gas intersect two isothermal curves as shown in$P-V$ diagram. The relation between the ratio $\frac{V_a}{V_d}$ and the ratio $\frac{V_b}{V_c}$ is:View Solution
- 2Unit mass of a liquid with volume ${V_1}$ is completely changed into a gas of volume ${V_2}$ at a constant external pressure $P$ and temperature $T.$ If the latent heat of evaporation for the given mass is $L,$ then the increase in the internal energy of the system isView Solution
- 3One mole of a monatomic ideal gas undergoes an adiabatic expansion in which its volume becomes eight times its initial value. If the initial temperature of the gas is $100 K$ and the universal gas constant $R =8.0 Jmol ^{-1} K ^{-1}$, the decrease in its internal energy, in Joule, is. . . . .View Solution
- 4When $1\, kg$ of ice at $0^o C$ melts to water at $0^o C,$ the resulting change in its entropy, taking latent heat of ice to be $80\, cal/gm,$ is ...... $cal/K$View Solution
- 5View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
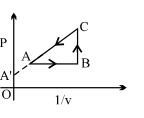
- 6A real gas within a closed chamber at $27^{\circ} \mathrm{C}$ undergoes the cyclic process as shown in figure. The gas obeys $P V^3=\mathrm{RT}$ equation for the path $A$ to $B$. The net work done in the complete cycle is (assuming $R=8 \mathrm{~J} / \mathrm{molK}$ ):View Solution
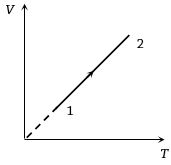
- 7Volume versus temperature graph of two moles of helium gas is as shown in figure. The ratio of heat absorbed and the work done by the gas in process $1-2$ isView Solution
- 8View Solution“Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
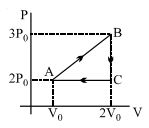
- 9View SolutionIn the above thermodynamic process, the correct statement is
- 10In a heat engine, the temperature of the source and sink are $500\, K$ and $375\, K$. If the engine consumes $25\times10^5\, J$ per cycle, the work done per cycle isView Solution
