An ideal monoatomic gas with pressure $P$, volume $V$ and temperature $T$ is expanded isothermally to a volume $2\, V$ and a final pressure $P_i$. If the same gas is expanded adiabatically to a volume $2\,V$, the final pressure is $P_a$ . The ratio $\frac{{{P_a}}}{{{P_i}}}$ is
AIEEE 2012, Medium
For isothermal process:
$P V=P_{t}, 2 V$
$P=2 P_{i}\,\,\,\,\,\,\,\,...(i)$
For adiabatic process $\mathrm{PV}^{\gamma}=\mathrm{P}_{\mathrm{a}}(2 \mathrm{V})^{\gamma}$
$(\because \text { for monatomic gas } \gamma=5 / 3)$
or, $\quad 2 \mathrm{P}_{\mathrm{i}} \mathrm{V}^{\frac{5}{3}}=P_{a}(2 \mathrm{V})^{\frac{5}{3}} \quad[\text { From }(\mathrm{i})]$
$\Rightarrow \frac{P_{a}}{P_{i}}=\frac{2}{2^{\frac{5}{3}}}$
$\Rightarrow \quad \frac{P_{a}}{P_i}=2^{\frac{-2}{3}}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
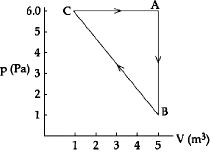
- 1For the given cyclic process $CAB$ as shown for a gas, the work done is ..... $J$View Solution
- 2View SolutionIn a thermodynamic system working substance is ideal gas, its internal energy is in the form of
- 3A system performs work $\Delta W$ when an amount of heat is$\Delta Q$ added to the system, the corresponding change in the internal energy is $\Delta U$. A unique function of the initial and final states (irrespective of the mode of change) isView Solution
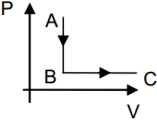
- 4If one mole of an ideal gas goes through the process $A \rightarrow B$ and $B \rightarrow C .$ Given that $T _{ A }=400\, K ,$ and $T _{ C }=400 \,K .$ If $\frac{ P _{ B }}{ P _{ A }}=\frac{1}{5},$ then find the heat supplied to the gas (in $J$)View Solution
- 5A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an abosolute temperature $T$ and rejects heat to a sink at a temperature of $T/3.$ The amount of heat rejected isView Solution
- 6Two moles of monoatomic gas is expanded from $(P_0, V_0)$ to $(P_0 , 2V_0)$ under isobaric condition. Let $\Delta Q_1$, be the heat given to the gas, $\Delta W_1$ the work done by the gas and $\Delta U_1$ the change in internal energy. Now the monoatomic gas is replaced by a diatomic gas. Other conditions remaining the same. The corresponding values in this case are $\Delta Q_2 , \Delta W_2 , \Delta U_2$ respectively, thenView Solution
- 7An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1 g/sec$. The calorific value of the fuel is $2 kcal/g$. Is the claim of the engineerView Solution
- 8Even Carnot engine cannot give $100\%$ efficiency because we cannotView Solution
- 9When an ideal gas $(\gamma = 5/3$) is heated under constant pressure, then what percentage of given heat energy will be utilised in doing external workView Solution
- 10A given ideal gas with $\gamma = \frac{{{C_p}}}{{{C_v}}} = 1.5$ at a temperature $T$. If the gas is compressed adiabatically to one-fourth of its initial volume, the final temperature will be ..... $T$View Solution
