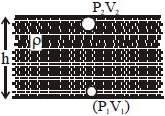
An inverted bell lying at the bottom of a lake $47.6\,m$ deep has $50\, cm^3$ of air trapped in it. The bell is brought to the surface of the lake. The volume of the trapped air will be ...... $cm^3$ (atmospheric pressure $= 70\, cm$ of $Hg$ and density of $Hg = 13.6\, g/cm^3$)
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The temperature of an ideal gas is increased from $200\,K$ to $800\,K$. If r.m.s. speed of gas at $200\,K$ is $v_0$. Then, r.m.s. speed of the gas at $800\,K$ will be:View Solution
- 2A jar contains a gas and few drops of water at $T K.$ The pressure in the jar is $830 \,mm$ of mercury. The temperature of jar is reduced by $1\%.$ The saturated vapour pressure of water at the two temperatures are $30 \,mm$ and $ 25\, mm$ of mercury. Then the new pressure in the jar will be ..... $mm$ of $Hg$View Solution
- 3If the $r.m.s.$ velocity of a gas at a given temperature (Kelvin scale) is $300\, m/sec$. What will be the $r.m.s.$ velocity of a gas having twice the molecular weight and half the temperature on Kelvin scale ....... $m/sec$View Solution
- 4View SolutionFor Boyle's law to hold the gas should be
- 5View SolutionA container with rigid walls is covered with perfectly insulating material. The container is divided into two parts by a partition. One part contains a gas while the other is fully evacuated (vacuum). The partition is suddenly removed. The gas rushes to fill the entire volume and comes to equilibrium after a little time. If the gas is not ideal, then
- 6One kg of a diatomic gas is at a pressure of $8 × 10^4\ N/m^2$. The density of the gas is $4\ kg/m^3$. What is the energy of the gas due to its thermal motion ?View Solution
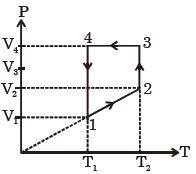
- 7A monoatomic ideal gas of two moles is taken through a cyclic process starting from $1$ as shown. $\frac{{{V_2}}}{{{V_1}}} = 2$ and $\frac{{{V_4}}}{{{V_1}}} = 4$ and temperature iast : $1$ is $T_1 = 27\,^oC$. The temperature at $2$ i.e., $T_2$ ...... $K$View Solution
- 8$14 \,g$ of $CO$ at $27^{\circ} C$ is mixed with $16 g$ of $O _2$ at $47^{\circ} C$. The temperature of mixture is .......... $^{\circ} C$ (vibration mode neglected)View Solution
- 9For a gas the difference between the two specific heats is $4150\, J/kg\, K.$ What is the specific heats at constant volume of gas if the ratio of specific heat is $1.4$View Solution
- 10A diatomic gas follows equation $PV^m =$ constant, during a process. What should be the value of $m$ such that its molar heat capacity during process $= R$View Solution