For Boyle's law to hold the gas should be
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At which temperature the r.m.s. velocity of a hydrogen molecule equal to that of an oxygen molecule at $47^{\circ} \mathrm{C}$ ?View Solution
- 2$STATEMENT- 1$ The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5$ times the product of its pressure and its volume. becauseView Solution
$STATEMENT-2$ The molecules of a gas collide with each other and the velocities of the molecules change due to the collision.
- 3At $10^o C$ the value of the density of a fixed mass of an ideal gas divided by it pressure is $x.$ At $110^o C$ this ratio isView Solution
- 4$105$ calories of heat is required to raise the temperature of $3$ moles of an ideal gas at constant pressure from $30^{\circ} C$ to $35^{\circ} C$. The amount of heat required in calories to raise the temperature of the gas through the range $\left(60^{\circ} C\right.$ to $\left.65^{\circ} C \right)$ at constant volume is ........ $cal$ $\left(\gamma=\frac{C_p}{C_v}=1.4\right)$View Solution
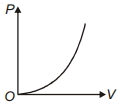
- 5The variation of pressure $P$ with volume $V$ for an ideal diatomic gas is parabolic as shown in the figure. The molar specific heat of the gas during this process isView Solution
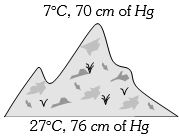
- 6At the top of a mountain a thermometer reads $7°C$ and a barometer reads $70\, cm$ of $Hg.$ At the bottom of the mountain these read $27°C$ and $76 \,cm$ of $Hg$ respectively. Comparison of density of air at the top with that of bottom isView Solution
- 7Consider a $1\, c.c.$ sample of air at absolute temperature ${T_0}$ at sea level and another $1 cc$ sample of air at a height where the pressure is one-third atmosphere. The absolute temperature $T$ of the sample at that height isView Solution
- 8If mass of $He$ atom is $4$ times that of hydrogen atom then mean velocity of $He$ isView Solution
- 9The $r.m.s.$ speed of the molecules of a gas in a vessel is $400$ $m{s^{ - 1}}$. If half of the gas leaks out, at constant temperature, the $r.m.s.$ speed of the remaining molecules will be ..... $ms^{-1}$View Solution
- 10An ideal gas of Molar mass $M$ is contained in a vertical tube of height $H$, closed at both ends. The tube is accelerating vertically upwards with acceleration $g$ Then, the ratio of pressure at the bottom and the mid point of the tube will beView Solution
