Assertion : A larger dry cell has higher $emf$
Reason : The $emf$ of a dry cell is proportional to its size
AIIMS 2004, Easy
The $e.m.f.$ of a dry cell is dependent upon the electrode potential of cathode and anode which in turn is dependent upon the reaction involved as well as concentration of the electrolyte. It has nothing to do with size of the cell. So, both assertion & reason are wrong.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a Wheatstone’s bridge all the four arms have equal resistance $R$. If the resistance of the galvanometer arm is also $R$, the equivalent resistance of the combination as seen by the battery isView Solution
- 2Dimensions of a block are $1\,cm \times 1\,cm \times 100\,cm$. If specific resistance of its material is $3 \times {10^{ - 7}}\,ohm - m$, then the resistance between the opposite rectangular faces isView Solution
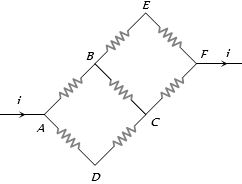
- 3In the adjoining circuit diagram each resistance is of $10$ $\Omega$. The current in the arm $AD$ will beView Solution
- 4The resistance of a conductor is $5\, ohm$ at $50\,^oC$ and $6\, ohm$ at $100\,^oC$. Its resistance at $0\,^oC$ is ................ $ohm$View Solution
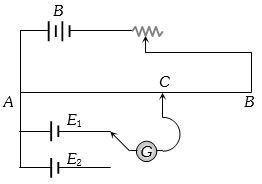
- 5The circuit shown here is used to compare the $e.m.f.$ of two cells ${E_1}$ and ${E_2}({E_1} > {E_2})$. The null point is at $C$ when the galvanometer is connected to ${E_1}$. When the galvanometer is connected to ${E_2}$, the null point will beView Solution
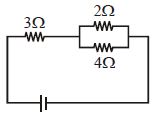
- 6If power in $3\,\Omega $ is $27\,W$ then what is the power in $2\,\Omega $ ................. $W$View Solution
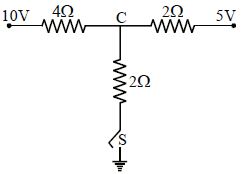
- 7As the switch $S$ is closed in the circuit shown in figure, current passed through it is .............View Solution
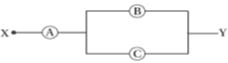
- 8$A, \,B$ and $C$ are voltmeters of resistance $R, \,1 .5R$ and $3R$ respectively as shown in the figure. When some potential difference is applied between $X$ and $Y,$ the voltmeter readings are $V_A, \,V_B$ and $V_C$ respectively. ThenView Solution
- 9The temperature dependence of resistances of $Cu$ and undoped $Si$ in the temperature range $300-400\ K$, is best described byView Solution
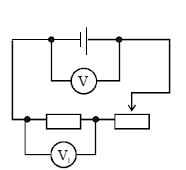
- 10If the rheostat slider were to move from the extreme right to the far left, How will the reading of voltmeter $V_1$ change?View Solution