$Assertion :$ Air quickly leaking out of a balloon becomes cooler.
$Reason :$ The leaking air undergoes adiabatic expansion.
$Reason :$ The leaking air undergoes adiabatic expansion.
AIIMS 2005, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In an adiabatic process, the state of a gas is changed from ${P_1},{V_1},{T_1} $ to ${P_2},{V_2},{T_2}$. Which of the following relation is correctView Solution
- 2View SolutionWhich of the following statements is correct for any thermodynamic system
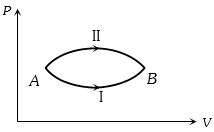
- 3A system goes from $A$ to $B$ via two processes $I$ and $II$ as shown in figure. If $\Delta {U_1}$ and $\Delta {U_2}$ are the changes in internal energies in the processes $I$ and $II$ respectively, thenView Solution
- 4View SolutionTwo gases have the same initial pressure, volume and temperatue. They expand to the same final volume, one adiabatically and the other isothermally
- 5Consider two containers $A$ and $B$ containing identical gases at the same pressure, volume and temperature. The gas in container $A$ is compressed to half of its original volume isothermally while the gas in container $B$ is compressed to half of its original value adiabatically. The ratio of final pressure of gas in $B$ to that of gas in $A$ isView Solution
- 6View Solution“Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
- 7The amount of work done in an adiabatic expansion from temperature $T$ to ${T_1}$ isView Solution
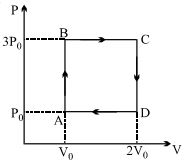
- 8A ideal monoatomic gas is carried around the cycle $ABCDA$ as shown in the fig. The efficiency of the gas cycle isView Solution
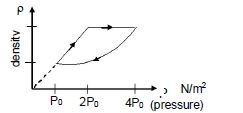
- 9An ideal gas undergoes cyclic process as shown in density pressure graph. During the process $AB$ the work done $|W_{AB}| = 70\,J$ . During the process $BC$, the gas absorbs $150\,J$ of heat. During the process $CA$ , gas undergoes expansion and does $210\,J$ of workView Solution
- 10View SolutionThe temperature of an ideal gas is kept constant as it expands. The gas does external work. During this process, the internal energy of the gas
