In an adiabatic process, the state of a gas is changed from ${P_1},{V_1},{T_1} $ to ${P_2},{V_2},{T_2}$. Which of the following relation is correct
Easy
(a) Adiabatic relation between $T$ and $V$ is
$T V_{1}^{\gamma-1}=$ constant
$\therefore \quad T_{1} V_{1}^{\gamma-1}=T_{2} V_{2}^{\gamma-1}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A Container having $1\ mole$ of a gas at a temperature $27\ ^oC$ has a movable piston which maintains at constant pressure in container of $1\ atm.$ The gas is compressed until temperature becomes $127^oC.$ The work done is ........ $J$ $(C_p\ for\ gas\ is\ 7.03\ cal/mol-K)$View Solution
- 2View SolutionThe slopes of isothermal and adiabatic curves are related as
- 3A van der Waal's gas obeys the equation of state $\left(p+\frac{n^2 a}{V^2}\right)(V-n b)=n R T$. Its internal energy is given by $U=C T-\frac{n^2 a}{V}$. The equation of a quasistatic adiabat for this gas is given byView Solution
- 4A gas expands adiabatically at constant pressure such that its temperature $T \propto \frac{1}{{\sqrt V }}$, the value of ${C_P}/{C_V}$ of gas isView Solution
- 5A thermo-dynamical system is changed from state $({P_1},\,{V_1})$ to $({P_2},\,{V_2})$ by two different process. The quantity which will remain same will beView Solution
- 6A gas at $NTP$ is suddenly compressed to one-fourth of its original volume. If $\gamma $ is supposed to be $\frac{3}{2}$, then the final pressure is........ atmosphereView Solution
- 7A sample of $0.1\, g$ of water at $100^o C$ and normal pressure $(1.013 \times 10^5 N m^{-2} )$ requires $54\ cal $ of heat energy to convert to steam at $100^o C.$ If the volume of the steam produced is $167.1 \,cc,$ the change in internal energy of the sample, is ....... $J$View Solution
- 8$Q$ amount of heat is given to $0.5\ mole$ of an ide al mono-atomic gas by a process $TV^n$ constant. Following graph shows variation of temperature with $Q$ . Find value of $n$.View Solution
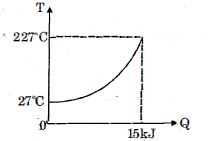
- 9A source supplies heat to a system at the rate of $1000 \,W$. If the system performs work at a rate of $200\,W$. The rate at which internal energy of the system increases $.......\,W$View Solution
- 10Consider two containers $A$ and $B$ containing identical gases at the same pressure, volume and temperature. The gas in container $A$ is compressed to half of its original volume isothermally while the gas in container $B$ is compressed to half of its original value adiabatically. The ratio of final pressure of gas in $B$ to that of gas in $A$ isView Solution
