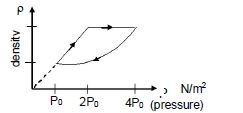
An ideal gas undergoes cyclic process as shown in density pressure graph. During the process $AB$ the work done $|W_{AB}| = 70\,J$ . During the process $BC$, the gas absorbs $150\,J$ of heat. During the process $CA$ , gas undergoes expansion and does $210\,J$ of work

Diffcult
| $Q$ | $\Delta U$ | $W$ | |
| $AB$ | $-70$ | $0$ | $-70$ |
| $BC$ | $150$ | $150$ | $0$ |
| $CA$ | $60$ | $-150$ | $210$ |
$\eta=\frac{140}{210} \times 100 \%=60 \%$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
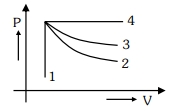
- 1An ideal gas undergoes four different processes from the same initial state as shown in the figure below. Those processes are adiabatic, isothermal, isobaric and isochoric. The curve which represents the adiabatic process among $1,2,3$ and $4$ isView Solution
- 2The isothermal Bulk modulus of an ideal gas at pressure $P$ isView Solution
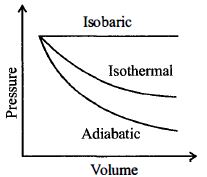
- 3A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?View Solution
- 4An insulator container contains $4\, moles$ of an ideal diatomic gas at temperature $T.$ Heat $Q$ is supplied to this gas, due to which $2 \,moles$ of the gas are dissociated into atoms but temperature of the gas remains constant. ThenView Solution
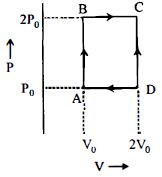
- 5An engine operates by taking $n\,moles$ of an ideal gas through the cycle $ABCDA$ shown in figure. The thermal efficiency of the engine is : (Take $C_v =1 .5\, R$, where $R$ is gas constant)View Solution
- 6Initial pressure and volume of a gas are $P$ and $V$ respectively. First it is expanded isothermally to volume $4V$ and then compressed adiabatically to volume $V$ . The final pressure of gas will be (given $\gamma = 3/2$ )View Solution
- 7A certain amount of gas of volume $V$ at $27^{o}\,C$ temperature and pressure $2 \times 10^{7} \;Nm ^{-2}$ expands isothermally until its volume gets doubled. Later it expands adiabatically until its volume gets redoubled. The final pressure of the gas will be (Use $\gamma=1.5$ )View Solution
- 8If a Carnot’s engine functions at source temperature $127^o C$ and at sink temperature $87^o C$, what is its efficiency ....... $\%$View Solution
- 9View SolutionIn an isothermal expansion
- 10A frictionless heat engine can be $100 \%$ efficient only if its exhaust temperature is ............View Solution
