$Assertion :$ In free expansion of an ideal gas, the entropy increases.
$Reason :$ Entropy increases in all natural processes.
$Reason :$ Entropy increases in all natural processes.
AIIMS 2007, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Determine efficiency of carnot cycle if in adiabatic expansion volume $3$ times of initial value and $\gamma =1.5$View Solution
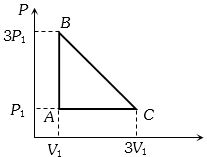
- 2An ideal gas is taken around the cycle $ABCA$ as shown in the $P-V $ diagram. The net work done by the gas during the cycle is equal toView Solution
- 3Water falls from a height of $60\,m$ at the rate of $15\,kg/s$ to operate a turbine. The losses due to frictional forces are $10\%$ of energy. How much power is generated by the turbine ....... $kW$ $(g = 10\,m/s^2)$View Solution
- 4In an adiabatic process, the density of a diatomic gas becomes $32$ times its initial value. The final pressure of the gas is found to be $n$ times the initial pressure. The value of $n$ isView Solution
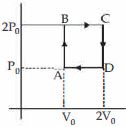
- 5Helium gas goes through a cycle $ABCDA$ ( consisting of two isochoric and isobaric lines) as shown in figure Efficiency of this cycle is nearly ....... $\%$ (Assume the gas to be close to ideal gas)View Solution
- 6In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 \,J$ of heat when $8 \,J$ of work was done on the gas. If the initial internal energy of the gas was $30 \,J$, then the final internal energy will be ........ $J$View Solution
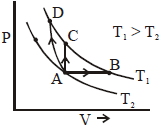
- 7Three different processes that can occur in an ideal monoatomic gas are shown in the $P$ vs $V$ diagram. The paths are labelled as $A \rightarrow B, A \rightarrow C$ and $A \rightarrow D .$ The change in internal energies during these process are taken as $E _{ AB }, E _{ AC }$ and $E _{ AD }$ and the work done as $W _{ AB }$ $W _{ AC }$ and $W _{ AD }$View Solution
The correct relation between these parameters are
- 8A mass of diatomic gas $(\gamma = 1 .4)$ at a pressure of $2$ atmospheres is compressed adiabatically so that its temperature rises from $27^o C$ to $927^o C.$ The pressure of the gas in the final state is ...... $atm$View Solution
- 9The equation of state for a gas is given by $PV = nRT + \alpha V$, where $n$ is the number of moles and $\alpha $ is a positive constant. The initial temperature and pressure of one mole of the gas contained in a cylinder are $T_o$ and $P_o$ respectively. The work done by the gas when its temperature doubles isobarically will beView Solution
- 10One mole of ${O_2}$ gas having a volume equal to $22.4$ litres at ${0^o}C$ and $1$ atmospheric pressure in compressed isothermally so that its volume reduces to $11.2$ litres. The work done in this process is ...... $J$View Solution
