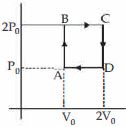
Helium gas goes through a cycle $ABCDA$ ( consisting of two isochoric and isobaric lines) as shown in figure Efficiency of this cycle is nearly ....... $\%$ (Assume the gas to be close to ideal gas)

AIEEE 2012, Diffcult
$Heat\,given\,to\,system = {\left( {n{C_V}\Delta T} \right)_{A \to B}} + {\left( {n{C_p}\Delta T} \right)_{B \to C}}$
$ = {\left[ {\frac{3}{2}\left( {nR\Delta T} \right)} \right]_{A \to B}} + {\left[ {\frac{5}{2}\left( {nR\Delta T} \right)} \right]_{B \to C}}$
$ = {\left[ {\frac{3}{2} \times {V_0}\Delta p} \right]_{A \to B}} + \left[ {\frac{5}{2} \times 2{P_0} \times {V_0}} \right]$
$ = \frac{{13}}{2}{P_0}{V_0}$
$and\,\,{W_0} = {P_0}{V_0}$
$\eta = \frac{{Work}}{{heat\,given}} = \frac{{{P_0}{V_0}}}{{\frac{{13}}{2}{P_0}{V_0}}} \times 100 = 15.4\% $
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A gas undergoes a change of state during which $100 J$ of heat is supplied to it and it does $20 J$ of work. The system is brought back to its original state through a process during which $20 J$ of heat is released by the gas. The work done by the gas in the second process is ....... $J$View Solution
- 2A Carnot's heat engine works between the temperatures $427^{\circ} C$ and $27^{\circ} C$. $...........\,kcal / s$ amount of heat should it consume per second to deliver mechanical work at the rate of $1.0\,kW$View Solution
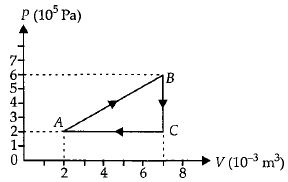
- 3A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
- 4Two gases of equal mass are in thermal equilibrium. If ${P_a},\,{P_b}$ and ${V_a}$ and ${V_b}$ are their respective pressures and volumes, then which relation is trueView Solution
- 5The efficiency of Carnot's engine operating between reservoirs, maintained at temperatures ${27^o}C$ and $ - {123^o}C,$ is ...... $\%$View Solution
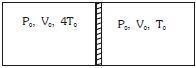
- 6Figure shows a cylindrical adiabatic container of total volume $2V_0$ divided into two equal parts by a conducting piston (which is free to move). Each part containing identical gas at pressure $P_0$ . Initially temperature of left and right part is $4T_0$ and $T_0$ respectively. An external force is applied on the piston to keep the piston at rest. Find the value of external force required when thermal equilibrium is reached. ( $A =$ Area of piston)View Solution
- 7A gas at initial temperature $T$ undergoes sudden expansion from volume $V$ to $2 \,V$. Then,View Solution
- 8An ideal Carnot engine, whose efficiency is $40 \%$ receives heat at $500\; K$. If its efficiency is $50 \%$ then the intake temperature for the same exhaust temperature is ......... $K$View Solution
- 9A Carnot's engine used first an ideal monoatomic gas then an ideal diatomic gas. If the source and sink temperature are ${411^o}C$ and ${69^o}C$ respectively and the engine extracts $1000\, J $ of heat in each cycle, then area enclosed by the $PV$ diagram is ........ $J$View Solution
- 10An ideal gas of mass $m$ in a state $A$ goes to another state $B$ via three different processes as shown in figure. If $Q_{1}, Q_{2}$ and $Q_{3}$ denote the heat absorbed by the gas along the three paths, thenView Solution