Determine efficiency of carnot cycle if in adiabatic expansion volume $3$ times of initial value and $\gamma =1.5$
AIIMS 2019, Medium
Consider the following expression.
$T _{1} V _{1}^{ \gamma -1}= T _{2} V _{2}^{ \gamma -1}$
$\frac{ T _{2}}{ T _{1}}=\left(\frac{ V _{1}}{ V _{2}}\right)^{ \gamma -1}$
Substitute the values.
$\frac{ T _{2}}{ T _{1}}=\left(\frac{1}{3}\right)^{1.5-1}$
$=\frac{1}{\sqrt{3}}$
The efficiency is given as,
$\eta=1-\frac{ T _{2}}{ T _{1}}$
$=1-\frac{1}{\sqrt{3}}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
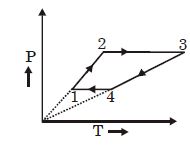
- 1Three moles of an ideal monoatomic gas perform a cycle as shown in the figure. The gas temperature in different states are: $T_1 = 400\, K, T_2 = 800\, K, T_3 = 2400\,K$ and $T_4 = 1200\,K.$ The work done by the gas during the cycle is ........ $kJ$View Solution
- 2Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the systemView Solution
Process Condition $(I)$ Adiabatic $(A)\; \Delta W =0$ $(II)$ Isothermal $(B)\; \Delta Q=0$ $(III)$ Isochoric $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ $(IV)$ Isobaric $(D)\; \Delta U =0$ - 3The volume of $1\; mole$ of an ideal gas with the adiabatic exponent $\gamma$ is changed according to the relation $V=\frac bT$ where $b =$ constant. The amount of heat absorbed by the gas in the process if the temperature is increased by $\triangle T$ will beView Solution
- 4A cylinder fitted with a piston contains $0.2 \,moles$ of air at temperature $27°C.$ The piston is pushed so slowly that the air within the cylinder remains in thermal equilibrium with the surroundings. Find the approximate work done by the system if the final volume is twice the initial volume ...... $J$View Solution
- 5A diatomic ideal gas is used in a Carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\ V$, the efficiency of the engine isView Solution
- 6The heat energy required to raise the temperature of $5\,moles$ of an ideal gas to $5\,K$ at constant pressure is $600\,J$ . How much heat (in $J$ ) is required to raise the same mass of the same gas to $5\,K$ at constant volume ? (Take $R = 8.3\,J/mole-^oK$ )View Solution
- 7An electric appliance supplies $6000\, {J} / {min}$ heat to the system. If the system delivers a power of $90\, {W}$. How long (in $sec$) it would take to increase the internal energy by $2.5 \times 10^{3}\, {J}$ ?View Solution
- 8An engine has an efficiency of $1/6.$ When the temperature of sink is reduced by $62^o C,$ its efficiency is doubled. Temperatures of the source is ....... $^oC$View Solution
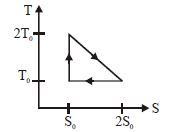
- 9View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
- 10One mole of ideal gas undergoes a linear process as shown in the figure below. Its temperature expressed as a function of volume $V$ isView Solution