At absolute zero temperature, pressure of a gas will be
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The average speed $v$ and $r.m.s.$ speed $\bar v$ of the molecules are related asView Solution
- 2View SolutionYou have two containers of equal volume. One is full of helium gas. The other holds an equal mass of nitrogen gas. Both gases have the same pressure. How does the temperature of the helium compare to the temperature of the nitrogen ?
- 3View SolutionThe absolute temperature of a gas is determined by
- 4Gas at a pressure ${P_0}$ in contained is a vessel. If the masses of all the molecules are halved and their speeds are doubled, the resulting pressure $P$ will be equal toView Solution
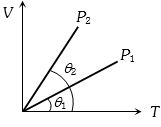
- 5The figure shows the volume $V$ versus temperature $T$ graphs for a certain mass of a perfect gas at two constant pressures of ${P_1}$ and ${P_2}$. What interference can you draw from the graphsView Solution
- 6Consider a mixture of $n$ moles of helium gas and $2 n$ moles of oxygen gas (molecules taken to be rigid) as an ideal gas. Its $\frac{\mathrm{C}_{\mathrm{P}}}{\mathrm{C}_{\mathrm{V}}}$ value will beView Solution
- 7The temperature at which the kinetic energy of oxygen molecules becomes double than its value at $27^{\circ}\,C$ is $............^{\circ}\,C$View Solution
- 8Ideal monoatomic gas is taken through a process $dQ = 2dU.$ The molar heat capacity for the processView Solution
- 9View SolutionA triatomic, diatomic and monatomic gas is supplied same amount of heat at constant pressure, then
- 10A gas is filled in a vessel at a pressure given by $P = \left( {6.02 \times {{10}^{23}}} \right)kT$ where $k$ is the Boltzmann constant and $T$ is the absolute temperature. The number of molecules per unit volume of the gas isView Solution
