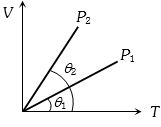
The figure shows the volume $V$ versus temperature $T$ graphs for a certain mass of a perfect gas at two constant pressures of ${P_1}$ and ${P_2}$. What interference can you draw from the graphs

IIT 1982,AIIMS 2014, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The temperature of an open room of volume $30\ m^3$ increases from $17^o C$ to $27vC$ due to sunshine. The atmospheric pressure in the room remains $1 \times 10^5\ Pa$. Ifni and nr are the number of molecules in the room before and after heating, then $n_f-n_i$ will be :View Solution
- 2The root mean square speed of the molecules of a diatomic gas is $v$. When the temperature is doubled, the molecules dissociate into two atoms. The new root mean square speed of the atom isView Solution
- 3The r.m.s. speed of a group of $7$ gas molecules having speeds $(6, 4, 2, 0, -2, -4, -6)\, m/s$ is ...... $m/s$View Solution
- 4For $a$ gas $C_{p}-C_{V}=R$ in a state $P$ and $C_{p}-C_{V}=1.10 R$ in a state $Q, T_{p}$ and $T_{Q}$ are the temperatures in two different states $P$ and $Q$ respectively. ThenView Solution
- 5The value of universal gas constant is $8.3\, J/mole/K,$ the mean kinetic energy of $32\, gm$ of oxygen at $-73°C$ will be ...... $J$View Solution
- 6View SolutionFor an ideal gas of diatomic molecules
- 7The $r.m.s.$ speed of the molecules of a gas in a vessel is $400$ $m{s^{ - 1}}$. If half of the gas leaks out, at constant temperature, the $r.m.s.$ speed of the remaining molecules will be ..... $ms^{-1}$View Solution
- 8A flask is filled with $13\, gm$ of an ideal gas at ${27}^o C$ and its temperature is raised to ${52}^o C$. The mass of the gas that has to be released to maintain the temperature of the gas in the flask at ${52}^o C$ and the pressure remaining the same is ..... $g$View Solution
- 9View SolutionAn ant is moving on a plane horizontal surface. The number of degrees of freedom of the ant will be .........
- 10When temperature of an ideal gas is increased from $27^\circ C$ to $227^\circ C$, its $r.m.s.$ speed changed from $400\, metre/sec$ to ${V_s}.$ The ${V_s}$ is ........ $metre/sec$View Solution
