You have two containers of equal volume. One is full of helium gas. The other holds an equal mass of nitrogen gas. Both gases have the same pressure. How does the temperature of the helium compare to the temperature of the nitrogen ?
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A mixture of ideal gases $7\,\, kg$ of nitrogen and $11 \,\,kg$ of $CO_2$. ThenView Solution
(Take $\gamma$ for nitrogen and $CO_2$ as $1.4$ and $1.3$ respectively)
- 2In Vander Waal's equation $\left[ {P + \frac{a}{{{V^2}}}} \right]\,(V - b) = RT,$ the dimensions of $a$ areView Solution
- 3The molar specific heat at constant pressure of an ideal gas is $(7/2)R.$ The ratio of specific heat at constant pressure to that at constant volume isView Solution
- 4If the ratio of vapour density for hydrogen and oxygen is $\frac{1}{{16}}$, then under constant pressure the ratio of their rms velocities will beView Solution
- 5The pressure $P$ of an ideal diatomic gas varies with its absolute temperature $T$ as shown in figure. The molar heat capacity of gas during this process is ........... $R$ [$R$ is gas constant]View Solution
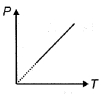
- 6Given below are two statements:View Solution
Statements $I:$ The temperature of a gas is $-73^{\circ}\,C$. When the gas is heated to $527^{\circ}\,C$, the root mean square speed of the molecules is doubled.
Statement $II:$ The product of pressure and volume of an ideal gas will be equal to translational kinetic energy of the molecules.
In the light of the above statements, choose the correct answer from the options given below :
- 7An ideal gas filled in a cylinder occupies volume $V$. The gas is compressed isothermally to the volume $V/3$. Now, the cylinder valve is opened and the gas is allowed to leak keeping temperature same. What percentage of the number of molecules should escape to bring the pressure in the cylinder back to its original value?View Solution
- 8The temperature of an ideal gas is increased from $120\, K$ to $480\, K.$ If at $120\, K,$ the root mean square velocity of the gas molecules is $v,$ at $480\, K$ it becomesView Solution
- 9For an ideal gas the instantaneous change in pressure $'{p} '$ with volume $'v'$ is given by the equation $\frac{{dp}}{{dv}}=-{ap} .$ If ${p}={p}_{0}$ at ${v}=0$ is the given boundary condition, then the maximum temperature one mole of gas can attain is :View Solution
(Here ${R}$ is the gas constant)
- 10View SolutionThree vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains helium (monoatomic), the second contains fluorine (diatomic) and the third contains sulfur hexafluoride (polyatomic). The correct statement, among the following is
