at constant pressure $Q=n C_{p} \Delta T$
$\Delta \mathrm{U}=\mathrm{nC}_{\mathrm{v}} \Delta \mathrm{T}$
$\Rightarrow \frac{W}{Q}=\frac{n \Delta T\left(C_{p}-C_{V}\right)}{n C_{p} \Delta T}=\frac{C_{p}-C_{V}}{C_{p}}=\frac{\gamma}{\gamma-1}$
Download our appand get started for free
Similar Questions
- 1Consider two containers $A$ and $B$ containing monoatomic gases at the same Pressure $(P)$, Volume $(V)$ and Temperature $(T)$. The gas in $A$ is compressed isothermally to $\frac{1}{8}$ of its original volume while the gas $B$ is compressed adiabatically to $\frac{1}{8}$ of its original volume. The ratio of final pressure of gas in $B$ to that of gas in $A$ is ...........View Solution
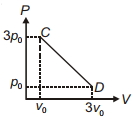
- 2The process $C D$ is shown in the diagram. As system is taken from $C$ to $D$, what happens to the temperature of the system?View Solution
- 3An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1\,g\,{s^{ - 1}}$. The calorific value of fuel is $2k cal/g$. His claimView Solution
- 4View SolutionAn ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
- 5One mole of an ideal gas at $300 \mathrm{~K}$ in thermal contact with surroundings expands isothermally from $1.0 \mathrm{~L}$ to $2.0 \mathrm{~L}$ against a constant pressure of $3.0 \mathrm{~atm}$. In this process, the change in entropy of surroundings $\left(\Delta S_{\text {surr }}\right)$ in $\mathrm{J} \mathrm{K}^{-1}$ is $(1 \mathrm{~L} \mathrm{~atm}=101.3 \mathrm{~J})$View Solution
- 6An engine is supposed to operate between two reservoirs at temperature $727°C$ and $227°C.$ The maximum possible efficiency of such an engine isView Solution
- 7A mixture of ideal gas containing $5$ moles of monatomic gas and $1$ mole of rigid diatomic gas is initially at pressure $P _0$, volume $V _0$ and temperature $T _0$. If the gas mixture is adiabatically compressed to a volume $V _0 / 4$, then the correct statement(s) is/are,View Solution
(Give $2^{1.2}=2.3 ; 2^{3.2}=9.2 ; R$ is gas constant)
$(1)$ The final pressure of the gas mixture after compression is in between $9 P _0$ and $10 P _0$
$(2)$ The average kinetic energy of the gas mixture after compression is in between $18 RT _0$ and $19 RT _0$
$(3)$ The work $| W |$ done during the process is $13 RT _0$
$(4)$ Adiabatic constant of the gas mixture is $1.6$
- 8A polyatomic gas with six degrees of freedom does $25\,\,J$ of work when it is expanded at constant pressure. The heat given to the gas is ..... $J$View Solution
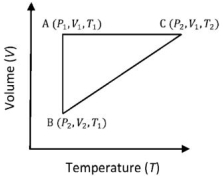
- 9A reversible cyclic process for an ideal gas is shown below. Here, $P , V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$ $(B)$ $W _{ BC }= P _2\left( V _2- V _1\right)$ and $q _{ BC }= H _{ AC }$ $(C)$ $\Delta H _{ CA }<\Delta U _{ CA }$ and $q _{ AC }=\Delta U _{ BC }$ $(D)$ $q_{B C}=\Delta H_{A C}$ and $\Delta H_{C A}>\Delta U_{C A}$
- 10$Assertion :$ Adiabatic expansion is always accompanied by fall in temperature.View Solution
$Reason :$ In adiabatic process, volume is inversely proportional to temperature.
