An engine is supposed to operate between two reservoirs at temperature $727°C$ and $227°C.$ The maximum possible efficiency of such an engine is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An engine runs between a reservoir at temperature $200 \,K$ and a hot body which is initially at temperature of $600 \,K$. If the hot body cools down to a temperature of $400 \,K$ in the process, then the maximum amount of work that the engine can do (while working in a cycle) is (the heat capacity of the hot body is $1 \,J / K )$View Solution
- 2A sample of gas at temperature $T$ is adiabatically expanded to double its volume. The work done by the gas in the process is $\left(\right.$ given, $\left.\gamma=\frac{3}{2}\right)$ :View Solution
- 3View SolutionIn thermodynamics, heat and work are
- 4A cylinder fitted with a piston contains $0.2 \,moles$ of air at temperature $27°C.$ The piston is pushed so slowly that the air within the cylinder remains in thermal equilibrium with the surroundings. Find the approximate work done by the system if the final volume is twice the initial volume ...... $J$View Solution
- 5An ideal gas undergoes four different processes from the same initial state as shown in the figure below. Those processes are adiabatic, isothermal, isobaric and isochoric. The curve which represents the adiabatic process among $1,2,3$ and $4$ isView Solution
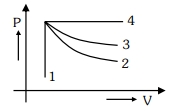
- 6A ideal monoatomic gas is carried around the cycle $ABCDA$ as shown in the fig. The efficiency of the gas cycle isView Solution
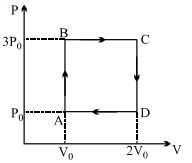
- 7At $N.T.P.$ one mole of diatomic gas is compressed adiabatically to half of its volume $\gamma = 1.41$. The work done on gas will be ....... $J$View Solution
- 8$Assertion :$ In adiabatic compression, the internal energy and temperature of the system get decreased.View Solution
$Reason :$ The adiabatic compression is a slow process. - 9The three processes in a thermodynamic cycle shown in the figure are : Process $1 \rightarrow 2$ is isothermal; Process $2 \rightarrow 3$ is isochoric (volume remains constant); Process $3 \rightarrow 1$ is adiabatic. The total work done by the ideal gas in this cycle is $10 \,J$. The internal energy decreases by $20 \,J$ in the isochoric process. The work done by the gas in the adiabatic process is $-20 \,J$. The heat added to the system in the isothermal process is .............. $J$View Solution
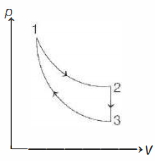
- 10A Carnot engine has efficiency $25\%$ . It operates between reservoirs of constant temperature with temperature difference of $80\,K$ . What is the temperature of low temperature reservoir ...... $^oC$View Solution
