At room temperature, the $r.m.s.$ speed of the molecules of certain diatomic gas is found to be $1920\, m/s$. The gas is
IIT 1984, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Five molecules of a gas are having speeds $1,2,4,8$ and $16\,m/s$ at some instant. The root mean square velocity of the gas molecules is ..... $m/s$View Solution
- 2The equation of state of a gas is given by $\left( {P + \frac{{a{T^2}}}{V}} \right)\,{V^c} = (RT + b)$, where $a, b, c$ and $R$ are constants. The isotherms can be represented by $P = A{V^m} - B{V^n}$, where $A$ and $B$ depend only on temperature thenView Solution
- 3View SolutionKinetic theory of gases was put forward by
- 4The correct relation between $\gamma=\frac{ C _p}{ c _v}$ and temperature $T$ is :View Solution
- 5View SolutionSelect the appropriate property of an ideal gas
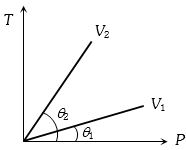
- 6View SolutionPressure versus temperature graphs of an ideal gas are as shown in figure. Choose the wrong statement
- 7View SolutionSelect the incorrect relation. (Where symbols have their usual meanings)
- 8If two moles of diatomic gas and one mole of mono atomic gas are mixed then the ratio of specific heats $\gamma=$ ......View Solution
- 9The relation between root mean square speed $\left( v _{ rms }\right)$ and most probable speed $\left( v _{ p }\right)$ for the molar mass $M$ of oxygen gas molecule at the temperature of $300\, K$ will beView Solution
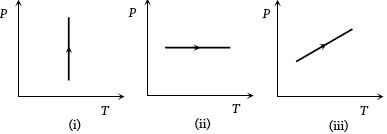
- 10From the following $P-T$ graph what interference can be drawnView Solution