At what temperature is the root mean square velocity of gaseous hydrogen molecules is equal to that of oxygen molecules at $47°C$ ..... $K$
AIEEE 2002, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
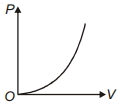
- 1The variation of pressure $P$ with volume $V$ for an ideal diatomic gas is parabolic as shown in the figure. The molar specific heat of the gas during this process isView Solution
- 2A container has $N$ molecules at absolute temperature $T$. If the number of molecules is doubled but kinetic energy in the box remain the same as before, the absolute temperature of the gas is ...........View Solution
- 3If $10^{22}$ gas molecules each of mass $10^{-26}\, kg$ collide with a surface (perpendicular to it)elastically per second over an area $1\, m^2$ with a speed $10^4\,m/s$, the pressure exerted by the gas molecules will be of the order ofView Solution
- 4View SolutionMolecules of a gas behave like
- 5What is the ratio of specific heats of constant pressure and constant volume for $N{H_3}$View Solution
- 6The temperature at which the average translational kinetic energy of a molecule is equal to the energy gained by an electron in accelerating from rest through a potential difference of $1\, volt$ isView Solution
- 7At $10^o C$ the value of the density of a fixed mass of an ideal gas divided by it pressure is $x.$ At $110^o C$ this ratio isView Solution
- 8The average kinetic energy of a gas molecule at $27^oC$ is $6.21 \times {10^{ - 21}}\,J$. Its average kinetic energy at $227°C$ will beView Solution
- 9A gas mixture consists of $3$ moles of oxygen and $5$ moles of argon at temperature $T$. Assuming the gases to be ideal and the oxygen bond to be rigid, the total internal energy (in units of $RT$ ) of the mixture isView Solution
- 10View SolutionA triatomic, diatomic and monatomic gas is supplied same amount of heat at constant pressure, then
