Avessel with open mouth contains air at $60^oC$. When the vessel is heated upto temperature $T$, one fourth of the air goes out. The value of $T$ is ..... $^oC$
Diffcult
$M_{1}=M, T_{1}=60+273$
$=333 K, M_{2}$
$=M-\frac{M}{4}=\frac{3 M}{4}$
[As $1/ 4$ th part of air escapes]
If pressure and volume of gas remains constant then $\mathrm{MT}=$ constant
$\therefore \frac{T_{2}}{T_{1}}=\frac{M_{1}}{M_{2}}=\left(\frac{M}{3 M / 4}\right)=\frac{4}{3} \Rightarrow T_{2}$
$=\frac{4}{3} \times T_{1}=\frac{4}{3} \times 333=444 \mathrm{K}$
$=171^{\circ} \mathrm{C}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider a spherical shell of radius $R$ at temperature $T$. The black body radiation inside it can be considered as an ideal gas of photons with internal energy per unit volume$E=$ $\frac{U}{V} \propto {T^4}$ and pressure $P = \frac{1}{3}\left( {\frac{U}{V}} \right)$ If the shell now undergoes an adiabatic expansion the relation between $T$ and $R$ isView Solution
- 2A sample of an ideal gas undergoes an isothermal expansion. If $d Q, d U$ and $d W$ represent the amount of heat supplied, the change in internal energy and the work done respectively, thenView Solution
- 3A monoatomic idea gas expands at constant pressure, with heat $Q$ supplied. The fraction of $Q$ which goes as work done by the gas isView Solution
- 4A Carnot's engine used first an ideal monoatomic gas then an ideal diatomic gas. If the source and sink temperature are ${411^o}C$ and ${69^o}C$ respectively and the engine extracts $1000\, J $ of heat in each cycle, then area enclosed by the $PV$ diagram is ........ $J$View Solution
- 5Air is pumped into a balloon, of initial volume $V$ , until its diameter has doubled. If the atmospheric pressure is $p$ , what is the work done against the atmosphere ?View Solution
- 6A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
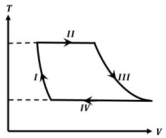
- 7One mole of a monatomic ideal gas undergoes a cyclic process as shown in the figure (where $V$ is the volume and $T$ is the temperature). Which of the statements below is (are) true?View Solution
(image)
$(A)$ Process $I$ is an isochoric process $(B)$ In process $II$, gas absorbs heat
$(C)$ In process $IV$, gas releases heat $(D)$ Processes $I$ and $III$ are $not$ isobaric
- 8Given that $1\,g$ of water in liquid phase has volume $1\,cm^3$ and in vapour phase $1671\, cm^3$ at atmospheric pressure and the latent heat of vaporization of water is $2256\,J/g;$ the change in the internal energy in joules for $1\,g$ of water at $373\,K$ when it changes from liquid phase to vapour phase at the same temperature is ....... $J$View Solution
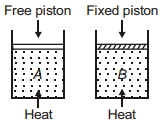
- 9Two cylinders contain same amount of ideal monatomic gas. Same amount of heat is given to two cylinders. If temperature rise in cylinder $A$ is $T_0$ then temperature rise in cylinder $B$ will be .........View Solution
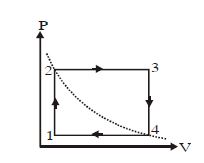
- 10One mole of an ideal gas undergoes a cyclic process, consisting of two isochores and two isobars. Temperature at $1$ and $3$ equal to $T_1$ and $T_3$ respectively. The work done by the gas over the cycle, if the point $2$ and $4$ lie on the same isothermView Solution