Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
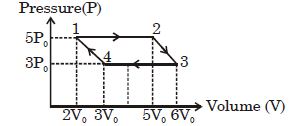
- 1An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?View Solution
- 2An ideal Carnot heat engine with an efficiency of $30\%$.It absorbs heat from a hot reservoir at $727^o C$. The temperature of the cold reservoir is .... $^oC$View Solution
- 3An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1\,g\,{s^{ - 1}}$. The calorific value of fuel is $2k cal/g$. His claimView Solution
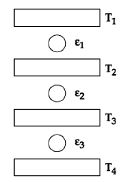
- 4Three Carnot engines operate in series between a heat source at a temperature $T_1$ and a heat sink at temperature $T_4$ (see figure). There are two other reservoirs at temperature $T_2$ and $T_3$, as shown, with $T_1 > T_2 > T_3 > T_4$. The three engines are equally efficient ifView Solution
- 5View SolutionWhich is the correct statement
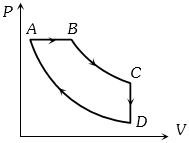
- 6A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same processView Solution
- 7A van der Waal's gas obeys the equation of state $\left(p+\frac{n^2 a}{V^2}\right)(V-n b)=n R T$. Its internal energy is given by $U=C T-\frac{n^2 a}{V}$. The equation of a quasistatic adiabat for this gas is given byView Solution
- 8Two Carnot engines $A$ and $B$ are operated in series. The first one, $A,$ receives heat at $T_1(= 600\,K)$ and rejects to a reservoir at temperature $T_2.$ The second engine $B$ receives heat rejected by the first engine and, in turns, rejects to a heat reservoir at $T_3 (=400\,K).$ Calculate the temperature $T_2$ if the work outputs of the two engines are equal ..... $K$View Solution
- 9View SolutionThe maximum possible efficiency of a heat engine is ...........
- 10Determine efficiency of carnot cycle if in adiabatic expansion volume $3$ times of initial value and $\gamma =1.5$View Solution
