Consider a gas with density $\rho $ and $\bar c$ as the root mean square velocity of its molecules contained in a volume. If the system moves as whole with velocity $v,$ then the pressure exerted by the gas is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
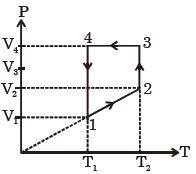
- 1A monoatomic ideal gas of two moles is taken through a cyclic process starting from $1$ as shown. $\frac{{{V_2}}}{{{V_1}}} = 2$ and $\frac{{{V_4}}}{{{V_1}}} = 4$ and temperature iast : $1$ is $T_1 = 27\,^oC$. The temperature at $2$ i.e., $T_2$ ...... $K$View Solution
- 2Consider two ideal diatomic gases $\mathrm{A}$ and $\mathrm{B}$ at some temperature $T$. Molecules of the gas $A$ are rigid, and have a mass $m$. Molecules of the gas $\mathrm{B}$ have an additional vibrational mode, and have a mass $\frac{\mathrm{m}}{4} .$ The ratio of the specific heats $(\mathrm{C}_{\mathrm{v}}^{\mathrm{A}}$ and $\mathrm{C}_{\mathrm{v}}^{\mathrm{B}})$ of gas $\mathrm{A}$ and $\mathrm{B}$, respectively isView Solution
- 3When one mole of monatomic gas is mixed with one mole of a diatomic gas, then the equivalent value of $\gamma$ for the mixture will be (vibration mode neglected)View Solution
- 4If three moles of monoatomic gas $\left(\gamma=\frac{5}{3}\right)$ is mixed with two moles of a diatomic gas $\left(\gamma=\frac{7}{5}\right)$, the value of adiabatic exponent $\gamma$ for the mixture is:View Solution
- 5At room temperature, the $r.m.s.$ speed of the molecules of certain diatomic gas is found to be $1920\, m/s$. The gas isView Solution
- 6In an ideal gas at temperature $T,$ the average force that a molecule applies on the walls of a closed container depends on $T$ as $T^q$ . A good estimate for $q$ isView Solution
- 7Relationship between $P,V,$ and $E$ for a gas isView Solution
- 8At a given temperature the ratio of $r.m.s.$ velocities of hydrogen molecule and helium atom will beView Solution
- 9A diatomic gas follows equation $PV^m =$ constant, during a process. What should be the value of $m$ such that its molar heat capacity during process $= R$View Solution
- 10View SolutionTwo vessels having equal volume contains molecular hydrogen at one atmosphere and helium at two atmospheres respectively. If both samples are at the same temperature, the mean velocity of hydrogen molecules is
