Two vessels having equal volume contains molecular hydrogen at one atmosphere and helium at two atmospheres respectively. If both samples are at the same temperature, the mean velocity of hydrogen molecules is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The lowest pressure (the best Vacuum) that can be created in the laboratory at $27$ degree is $10^{-11} \;{mm}$ of $Hg$. At this pressure, the number of ideal gas molecules per ${cm}^{3}$ will beView Solution
- 2View SolutionSelect the incorrect statement about the specific heats of a gaseous system.
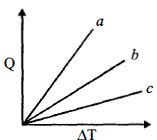
- 3Figure shows the variation in temperature $\left( {\Delta T} \right)$ with the amount of heat supplied $(Q)$ in an isobaric process corresponding to a monoatomic $(M)$, diatomic $(D)$ and a polyatomic $(P)$ gas. The initial state of all the gases are the same and the scales for the two axes coincide. Ignoring vibrational degrees of freedom, the lines $a, b$ and $c$ respectively correspond toView Solution
- 4View SolutionFor an ideal gas
- 5View SolutionAccording to kinetic theory of gases,
- 6A container $X$ has volume double that of contianer $Y$ and both are connected by a thin tube. Both contains same ideal gas. The temperature of $X$ is $200\,\,K$ and that of $Y$ is $400\,\,K$. If mass of gas in $X$ is $m$ then in $Y$ it will be:View Solution
- 7The temperature $(T)$ of one mole of an ideal gas varies with its volume $(V)$ as $T=-\alpha V^3+\beta V^2$, where $\alpha$ and $\beta$ are positive constants. The maximum pressure of gas during this process is ............View Solution
- 8At temperature $T,$ the $r.m.s.$ speed of helium molecules is the same as $r.m.s.$ speed of hydrogen molecules at normal temperature and pressure. The value of $T$ is ....... $^oC$View Solution
- 9View SolutionOn colliding in a closed container the gas molecules
- 10View SolutionThe specific heats at constant pressure is greater than that of the same gas at constant volume because
