Dehydration of the following in increasing order is:
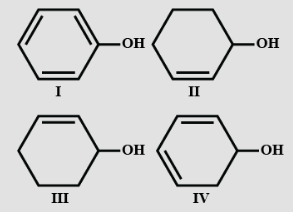

- I
Phenol (I) is stable aromatic compound. So it is most stable. The diene (IV) undergoes quick dehydration because it will form stable phenol. The alkene (III) undergoes dehydration. less readily because it gives a more stable diene. Alcohol (II) undergoes much lesser dehydration to form alkene. So, order is: 4>3>2>1.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionEthanol on reaction with acetic anhydride gives:
- 2View SolutionWhich of the following alkenes on acid-catalysed hydration gives a tertiary alcohol:
- 3The only alcohol that can be prepared by the indirect hydration of alkene is$:$View Solution
- 4View SolutionEthers show dipolar nature due to the presence of ______.
- 5$........$ is prepared on a large seals from water gas.View Solution
- 6View SolutionWhich of the following alcohol has the highest boiling point?
- 7The best method to prepare $3-$methylbutan$-2-ol$ from $3-$methylbut$-1-$ene is:View Solution
- 8View SolutionWhich of the following represents monohydric alcohol?
- 9View SolutionCommon name for methanol is:
- 10View SolutionWhich of the following is wood alcohol?
