The best method to prepare $3-$methylbutan$-2-ol$ from $3-$methylbut$-1-$ene is:
To prepare $3-$methylbutan$-2-ol$ from $3-$methylbut$-1-$ene shouls involve Markovnikov's addition of $H−OH$ across double bond.Addition of water in presence of dil. $\ce{H_2SO_4}$ follows Markovnikov's addition of $H−OH$ across the alkene.
Addition of $\text{HCI}$ followed by reaction with dil. $\text{NaOH}$ to an alkene can give substitution and elimination product after alkyl halide formation.
Hydroboration$-$oxidation reaction follows Anti$-$Markovnikov's addition of $H−OH.$
Reimer $-$ Tiemann reaction is a chemical reaction used for the ortho$-$formylation of phenols.
Thus the best method to prepare $3-$methylbutan$-2-ol$ from $3-$methylbut$-1-$ene is the addition of water in presence of dil. $\ce{H_{2}SO_4}.$
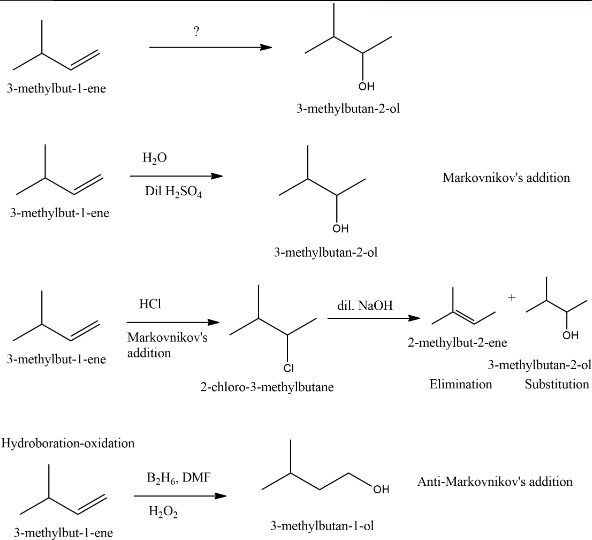
Addition of $\text{HCI}$ followed by reaction with dil. $\text{NaOH}$ to an alkene can give substitution and elimination product after alkyl halide formation.
Hydroboration$-$oxidation reaction follows Anti$-$Markovnikov's addition of $H−OH.$
Reimer $-$ Tiemann reaction is a chemical reaction used for the ortho$-$formylation of phenols.
Thus the best method to prepare $3-$methylbutan$-2-ol$ from $3-$methylbut$-1-$ene is the addition of water in presence of dil. $\ce{H_{2}SO_4}.$

Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Which of the following alcohols is dehydrated most easily with conc. $\ce{H_2SO_4}$?View Solution
- 2Consider the reaction between $\ce{C_2H_{5}O^{⊝ }}$ and dimethyl sulphate. The leaving group in this reaction is$:$View Solution
- 3View SolutionWhich of the following cannot be dissolved in alcohol?
- 4View SolutionThe correct IUPAC name of the compound is?

- 5View SolutionMark the correct statement:
- 6During dehydration of alcohols to alkenes by heating with conc. $\ce{H_2SO_{4}},$ the initiation step is$:$View Solution
- 7When $3-$methylbutan$-2-$ol is treated with $\ce{HBr},$ the following reaction takes place. What are the correct steps for formation of the above product?View Solution
- 8View SolutionThe IUPAC name of the following compound is:
- 9View SolutionWhat is the functional group in alcohol?
- 10View SolutionWhich of the following alcohol has the highest boiling point?
