Determine coefficient of performance of given temperature limit.
$T_{1}=27^{\circ} C$ [outside fridge]
$T_{2}=-23^{\circ} C$ [inside fridge]
AIIMS 2019, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The equation of state for a gas is given by $PV = nRT + \alpha V$, where $n$ is the number of moles and $\alpha $ is a positive constant. The initial temperature and pressure of one mole of the gas contained in a cylinder are $T_o$ and $P_o$ respectively. The work done by the gas when its temperature doubles isobarically will beView Solution
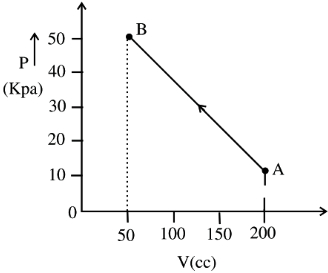
- 2The pressure of a gas changes linearly with volume from $A$ to $B$ as shown in figure. If no heat is supplied to or extracted from the gas then change in the internal energy of the gas will be $............\,J$View Solution
- 3At $N.T.P.$ one mole of diatomic gas is compressed adiabatically to half of its volume $\gamma = 1.41$. The work done on gas will be ....... $J$View Solution
- 4An engine takes in $5$ moles of air at $20\,^{\circ} C$ and $1$ $atm,$ and compresses it adiabaticaly to $1 / 10^{\text {th }}$ of the original volume. Assuming air to be a diatomic ideal gas made up of rigid molecules, the change in its internal energy during this process comes out to be $X\, kJ$. The value of $X$ to the nearest integer isView Solution
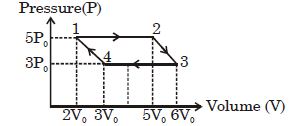
- 5An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?View Solution
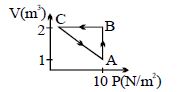
- 6An ideal gas is taken through the cycle $A \to B \to C \to A$ , as shown in the figure. If the net heat supplied to the gas in the cycle is $5\ J$, the work done by the gas in the process $C \to A$ is .... $J$View Solution
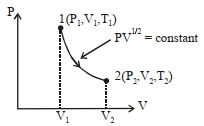
- 7Thermodynamic process is shown below on a $P-V$ diagram for one mole of an ideal gas. If $V _{2}=2 V _{1}$ then the ratio of temperature $T _{2} / T _{1}$ is ...... .View Solution
- 8A Carnot's engine used first an ideal monoatomic gas then an ideal diatomic gas. If the source and sink temperature are ${411^o}C$ and ${69^o}C$ respectively and the engine extracts $1000\, J $ of heat in each cycle, then area enclosed by the $PV$ diagram is ........ $J$View Solution
- 9View SolutionThe thermodynamic process, in which internal energy of the system remains constant is ...........
- 10If a Carnot engine works between $127^{\circ} C$ and $527^{\circ} C$, then its efficiency is ...... $\%$View Solution
