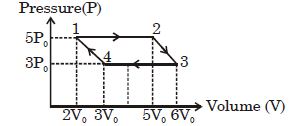
An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
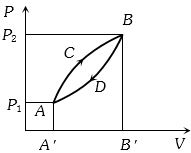
- 1A thermodynamic system is taken from state $A$ to $B$ along $ACB$ and is brought back to $A$ along $BDA$ as shown in the $PV$ diagram. The net work done during the complete cycle is given by the areaView Solution
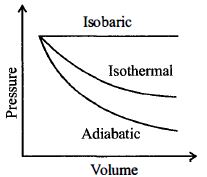
- 2If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution
- 3View SolutionThe maximum possible efficiency of a heat engine is ...........
- 4View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
- 5A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?View Solution
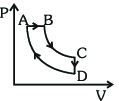
- 6A cyclic process $ABCDA$ is shown in the $P-V$ diagram. Which of the following curves represent the same processView Solution
- 7View SolutionMonoatomic, diatomic and triatomic gases whose initial volume and pressure are same, are compressed till their volume becomes half the initial volume.
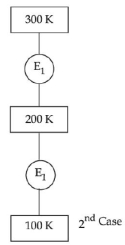
- 8In $1^{\text {st }}$ case, Carnot engine operates between temperatures $300\,K$ and $100\,K$. In $2^{\text {nd }}$ case, as shown in the figure, a combination of two engines is used. The efficiency of this combination (in $2^{\text {ad }}$ case) will be.View Solution
- 9During an isothermal expansion, a confined ideal gas does $-150 \,J$ of work against its surroundings. This implies thatView Solution
- 10$1\,kg$ of water at $100\, ^{\circ}C$ is converted into steam at $100^{\circ}\,C$ by boiling at atmospheric pressure. The volume of water changes from $1.00 \times 10^{-3}\,m ^3$ as a liquid to $1.671\,m ^3$ as steam. The change in internal energy of the system during the process will be $........kJ$ (Given latent heat of vaporisaiton $=2257\,kJ / kg$. Atmospheric pressure $=1 \times 10^5\,Pa$ )View Solution
