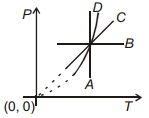
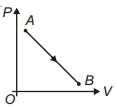
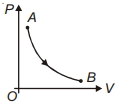
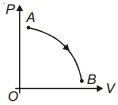
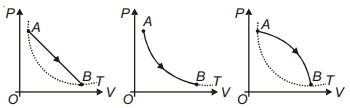
During which of the following thermodynamic process represented by $P V$ diagram the heat energy absorbed by system may be equal to area under $P V$ graph?
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A source supplies heat to a system at the rate of $1000 \,W$. If the system performs work at a rate of $200\,W$. The rate at which internal energy of the system increases $.......\,W$View Solution
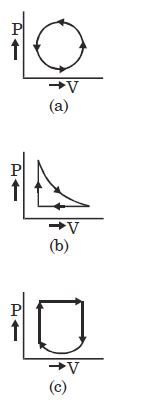
- 2View SolutionWhat is the nature of change in internal energy in the following three thermodynamical processes shown in figure
- 3One mole of a monoatomic ideal gas $\left(c_{ V }=\frac{3}{2} R \right)$ undergoes a cycle where it first goes isochorically from the state $\left(\frac{3}{2} P _0, V _0\right)$ to $\left( P _0, V _0\right)$, and then is isobarically contracted to the volume $\frac{1}{2} V _0$. It is then taken back to the initial state by a path which is a quarter ellipse on the $P - V$ diagram. The efficiency of this cycle isView Solution
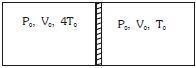
- 4Figure shows a cylindrical adiabatic container of total volume $2V_0$ divided into two equal parts by a conducting piston (which is free to move). Each part containing identical gas at pressure $P_0$ . Initially temperature of left and right part is $4T_0$ and $T_0$ respectively. An external force is applied on the piston to keep the piston at rest. Find the value of external force required when thermal equilibrium is reached. ( $A =$ Area of piston)View Solution
- 5When an ideal gas in a cylinder was compressed isothermally by a piston, the work done on the gas was found to be $1.5 \times {10^4}\;joules$. During this process aboutView Solution
- 6Two identical containers $A$ and $B$ with frictionless pistons contain the same ideal gas at the same temperature and the same volume $V$. The mass of the gas in $A$ is ${m_A}$ and that in $B$ is ${m_B}$. The gas in each cylinder is now allowed to expand isothermally to the same final volume $2V$. The changes in the pressure in $A$ and $B$ are found to be $\Delta P$ and $1.5 \Delta P$ respectively. ThenView Solution
- 7One mole of an ideal gas is taken through an adiabatic process where the temperature rises from $27^{\circ} {C}$ to $37^{\circ} {C}$. If the ideal gas is composed of polyatomic molecule that has $4$ vibrational modes which of the following is true?View Solution
- 8Two carnot engines $A$ and $B$ operate in series such that engine $A$ absorbs heat at $T_{1}$ and rejects heat to a sink at temperature $T$. Engine $B$ absorbs half of the heat rejected by engine $A$ and rejects heat to the sink at ${T}_{3}$. When workdone in both the cases is equal, the value of ${T}$ isView Solution
- 9View SolutionA gas is compressed adiabatically till its temperature is doubled. The ratio of its final volume to initial volume will be
- 10Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternative.View Solution