When an ideal gas in a cylinder was compressed isothermally by a piston, the work done on the gas was found to be $1.5 \times {10^4}\;joules$. During this process about
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionTwo gases are said to be in thermal equilibrium when they have same
- 2View SolutionFor which combination of working temperatures the efficiency of Carnot’s engine is highest
- 3View SolutionIn thermodynamics, heat and work are
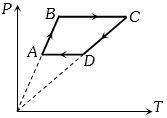
- 4Six moles of an ideal gas perfomrs a cycle shown in figure. If the temperature are $T_A = 600 K, T_B = 800 K, T_C = 2200 K \,and\, T_D = 1200 K$, the work done per cycle is ..... $kJ$View Solution
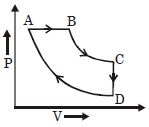
- 5A cyclic process $ABCD$ is shown in the given $P-V$ diagram. $P-T$ diagram that represents the same process isView Solution
- 6A container of volume $1{m^3}$is divided into two equal compartments by a partition. One of these compartments contains an ideal gas at $300 K$. The other compartment is vacuum. The whole system is thermally isolated from its surroundings. The partition is removed and the gas expands to occupy the whole volume of the container. Its temperature now would be ..... $K$View Solution
- 7A litre of dry air at $STP$ expands adiabatically to a volume of $3$ litres. If $\gamma=1.40,$ the work done by air is$(3^{1.4}=4.6555)$ [Take air to be an ideal gas $]$View Solution
- 8Water falls from a height of $60\,m$ at the rate of $15\,kg/s$ to operate a turbine. The losses due to frictional forces are $10\%$ of energy. How much power is generated by the turbine ....... $kW$ $(g = 10\,m/s^2)$View Solution
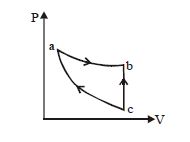
- 9An ideal gas expands isothermally along $ab$ and does $600\,J$ of work. During the processView Solution
- 10A system performs work $\Delta W$ when an amount of heat is$\Delta Q$ added to the system, the corresponding change in the internal energy is $\Delta U$. A unique function of the initial and final states (irrespective of the mode of change) isView Solution
