Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
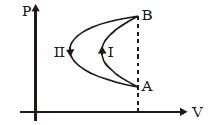
- 1In a cyclic process, a gas is taken from state $A$ to $B$ via path $-I$ as shown in the indicator diagram and taken back to state $A$ from state $B$ via path $-II$ . In the complete cycleView Solution
- 2For a refrigerator, heat absorbed from source is $800\, J$ and heat supplied to sink is $500\, J$ then the coefficient of performance isView Solution
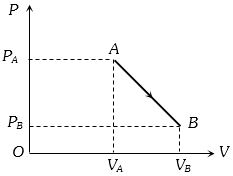
- 3An ideal gas is taken from point $A$ to the point $B,$ as shown in the $P-V$ diagram, keeping the temperature constant. The work done in the process isView Solution
- 4A rigid diatomic ideal gas undergoes an adiabatic process at room temperature. The rational between temperature and volume for the process is $TV^x =$ constant, then $x$ isView Solution
- 5Helium gas goes through a cycle $ABCDA$ ( consisting of two isochoric and isobaric lines) as shown in figure Efficiency of this cycle is nearly ....... $\%$ (Assume the gas to be close to ideal gas)View Solution
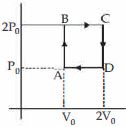
- 6The amount of work done in an adiabatic expansion from temperature $T$ to ${T_1}$ isView Solution
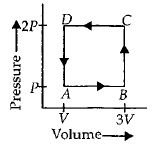
- 7A thermodynamic system is taken through the cycle $ABCD$ as shown in figure. Heat rejected by the gas during the cycle isView Solution
- 8An ideal heat engine exhausting heat at $77\,^oC$. To have a $30\%$ efficiency. It must take heat at...... $^oC$View Solution
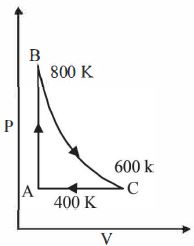
- 9One mole of a diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\ K, 800\ K $ and $600\ K$ respectively. Choose the correct statementView Solution
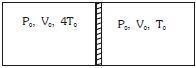
- 10Figure shows a cylindrical adiabatic container of total volume $2V_0$ divided into two equal parts by a conducting piston (which is free to move). Each part containing identical gas at pressure $P_0$ . Initially temperature of left and right part is $4T_0$ and $T_0$ respectively. An external force is applied on the piston to keep the piston at rest. Find the value of external force required when thermal equilibrium is reached. ( $A =$ Area of piston)View Solution