Entropy of a system decreases .........
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ Thermodynamic process in nature are irreversible.View Solution
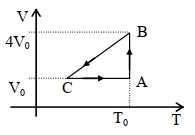
$Reason :$ Dissipative effects cannot be eliminated. - 2One mole of an ideal gas in initial state $\mathrm{A}$ undergoes a cyclic process $A B C A$, as shown in the figure. Its pressure at $A$ is $\mathrm{P}_0$. Choose the correct option$(s)$ from the followingView Solution
$(A)$ Internal energies at $\mathrm{A}$ and $\mathrm{B}$ are the same
$(B)$ Work done by the gas in process $\mathrm{AB}$ is $\mathrm{P}_0 \mathrm{~V}_0 \ln 4$
$(C)$ Pressure at $C$ is $\frac{P_0}{4}$
$(D)$ Temperature at $\mathrm{C}$ is $\frac{\mathrm{T}_0}{4}$
- 3View SolutionThe first operation involved in a Carnot cycle is
- 4One mole of an ideal gas at an initial temperature of $T\, K$ does $6 R$ joules of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $5/3$, the final temperature of gas will beView Solution
- 5In a heat engine, the temperature of the source and sink are $500\, K$ and $375\, K$. If the engine consumes $25\times10^5\, J$ per cycle, the work done per cycle isView Solution
- 6The pressure $P_{1}$ and density $d_{1}$ of diatomic gas $\left(\gamma=\frac{7}{5}\right)$ changes suddenly to $P _{2}\left(> P _{1}\right)$ and $d _{2}$ respectively during an adiabatic process. The temperature of the gas increases and becomes $......$ times of its initial temperature.$\left(\right.$ given $\left.\frac{ d _{2}}{ d _{1}}=32\right)$View Solution
- 7A Carnot engine whose heat $\operatorname{sinks}$ at $27\,^{\circ} C$, has an efficiency of $25 \%$. By how many degrees should the temperature of the source be changed to increase the efficiency by $100 \%$ of the original efficiency $?$View Solution
- 8A gas for which $\gamma = 1.5$ is suddenly compressed to $\frac{1}{4}$ th of the initial volume. Then the ratio of the final to the initial pressure isView Solution
- 9For adiabatic processes $\left( {\gamma = \frac{{{C_p}}}{{{C_v}}}} \right)$View Solution
- 10The efficiency of carnot engine is $50\%$ and temperature of sink is $500\;K$. If temperature of source is kept constant and its efficiency raised to $60\%$, then the required temperature of the sink will beView Solution
