The first operation involved in a Carnot cycle is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhen heat in given to a gas in an isobaric process, then
- 2An ideal monoatomic gas with pressure $P$, volume $V$ and temperature $T$ is expanded isothermally to a volume $2\, V$ and a final pressure $P_i$. If the same gas is expanded adiabatically to a volume $2\,V$, the final pressure is $P_a$ . The ratio $\frac{{{P_a}}}{{{P_i}}}$ isView Solution
- 3A Carnot's engine used first an ideal monoatomic gas then an ideal diatomic gas. If the source and sink temperature are ${411^o}C$ and ${69^o}C$ respectively and the engine extracts $1000\, J $ of heat in each cycle, then area enclosed by the $PV$ diagram is ........ $J$View Solution
- 4View SolutionIrreversible process is
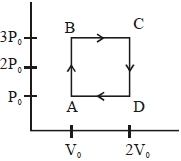
- 5An engine operates by taking a monatomic ideal gas through the cycle shown in the figure. The percentage efficiency of the engine is close to $.......\%$View Solution
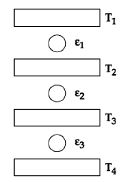
- 6Three Carnot engines operate in series between a heat source at a temperature $T_1$ and a heat sink at temperature $T_4$ (see figure). There are two other reservoirs at temperature $T_2$ and $T_3$, as shown, with $T_1 > T_2 > T_3 > T_4$. The three engines are equally efficient ifView Solution
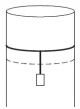
- 7A long cylindrical pipe of radius $20 \,cm$ is closed at its upper end and has an airtight piston of negligible mass as shown. When a $50 \,kg$ mass is attached to the other end of the piston, it moves down. If the air in the enclosure is cooled from temperature $T$ to $T-\Delta T$, the piston moves back to its original position. Then $\Delta T / T$ is close to (Assuming air to be an ideal gas, $g=10 \,m / s ^2$, atmospheric pressure is $10^5 \,Pa$ )View Solution
- 8A vessel containing $5\, litres$ of a gas at $0.8 \,pa$ pressure is connected to an evacuated vessel of volume $3$ litres. The resultant pressure inside will be ...... $pa$ (assuming whole system to be isolated)View Solution
- 9A heat engine operates between a cold reservoir at temperature ${T}_{2}=400\, {K}$ and a hot reservoir at temperature ${T}_{1} .$ It takes $300 \,{J}$ of heat from the hot reservoir and delivers $240\, {J}$ of heat to the cold reservoir in a cycle. The minimum temperature of the hot reservoir has to be $....{K}$View Solution
- 10An ideal gas heat engine operates in a Carnot cycle between $227^o C$ and $127^o C$. It absorbs $6\,kcal$ at the higher temperature. The amount of heat (in $kcal$) converted into work is equal toView Solution
