The efficiency of carnot engine is $50\%$ and temperature of sink is $500\;K$. If temperature of source is kept constant and its efficiency raised to $60\%$, then the required temperature of the sink will be
AIPMT 2002, Medium
(b) $\eta = 1 - \frac{{{T_2}}}{{{T_1}}}$==>$\frac{1}{2} = 1 - \frac{{500}}{{{T_1}}}$==> $\frac{{500}}{{{T_1}}} = \frac{1}{2}$…..$(i)$
$\frac{{60}}{{100}} = 1 - \frac{{{T_2}'}}{{{T_1}}}$==>$\frac{{{T_2}'}}{{{T_1}}} = \frac{2}{5}$…..$(ii)$
Dividing equation $(i)$ by $(ii)$, $\frac{{500}}{{{T_2}'}} = \frac{5}{4}$==>${T_2} = 400K$
$\frac{{60}}{{100}} = 1 - \frac{{{T_2}'}}{{{T_1}}}$==>$\frac{{{T_2}'}}{{{T_1}}} = \frac{2}{5}$…..$(ii)$
Dividing equation $(i)$ by $(ii)$, $\frac{{500}}{{{T_2}'}} = \frac{5}{4}$==>${T_2} = 400K$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$. The change in the internal energy of the gas is .... $J$ (given $R = 8.3 \,J/mole/K$)View Solution
- 2Which of the following is an equivalent cyclic process corresponding to the thermodynamic cyclic given in the figure? where, $1 \rightarrow 2$ is adiabatic.View Solution
(Graphs are schematic and are not to scale)
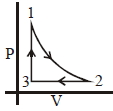
- 3An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution
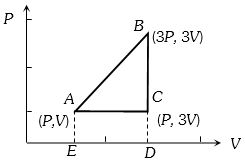
- 4In a thermodynamics process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20\,J$ of heat and $8\,J$ of work is done on the gas. If the initial internal energy of the gas was $30\,J$. The final internal energy will be...... $J$View Solution
- 5Two identical samples of a gas are allowed to expand $(i)$ isothermally $(ii)$ adiabatically. Work done isView Solution
- 6For a refrigerator, heat absorbed from source is $800\, J$ and heat supplied to sink is $500\, J$ then the coefficient of performance isView Solution
- 7An ideal refrigerator has a freezer at a temperature of $ - 13^\circ C.$ The coefficient of performance of the engine is $5.$ The temperature of the air (to which heat is rejected) will beView Solution
- 8If $\Delta U$ and $\Delta W$ represent the increase in internal energy and work done by the system respectively in a thermodynamical process, which of the following is true?View Solution
- 9A source supplies heat to a system at the rate of $1000 \,W$. If the system performs work at a rate of $200\,W$. The rate at which internal energy of the system increases $.......\,W$View Solution
- 10An ideal gas at a pressures of $1$ atmosphere and temperature of ${27^o}C$ is compressed adiabatically until its pressure becomes $8$ times the initial pressure, then the final temperature is ..... $^oC$ ($\gamma = 3/2$)View Solution
