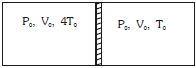
$\frac{\mathrm{P}_{0} \mathrm{V}_{0}}{\mathrm{R} 4 \mathrm{T}_{0}} \mathrm{C}_{\mathrm{V}}\left(4 \mathrm{T}_{0}-\mathrm{T}\right)=\frac{\mathrm{P}_{0} \mathrm{V}_{0}}{\mathrm{RT}_{0}} \mathrm{C}_{\mathrm{V}}\left(\mathrm{T}-\mathrm{T}_{0}\right)$
$\frac{4 \mathrm{T}_{0}-\mathrm{T}}{4}=\mathrm{T}-\mathrm{T}_{0}$
$4 \mathrm{T}_{0}-\mathrm{T}=4 \mathrm{T}-4 \mathrm{T}_{0}$
$8 \mathrm{T}_{0}=5 \mathrm{T}$
$\mathrm{T}=\frac{8 \mathrm{T}_{0}}{5}$
Final pressure
In left $\quad \mathrm{P}_{\mathrm{f}}=\frac{\mathrm{T}_{\mathrm{f}}}{\mathrm{T}_{0}} \mathrm{P}_{1}=\frac{\frac{8}{5} \mathrm{T}_{0}}{4 \mathrm{T}_{0}}=\frac{2}{5} \mathrm{P}_{0}$
In right $\quad \mathrm{P}_{\mathrm{f}}=\frac{\frac{8}{5} \mathrm{T}_{0}}{\mathrm{T}_{0}}=\mathrm{P}_{0}=\frac{8}{5} \mathrm{P}_{0}$
Force $=\left(\frac{8 \mathrm{P}_{0}}{5}-\frac{2 \mathrm{P}_{0}}{5}\right) \mathrm{A}=\frac{6 \mathrm{P}_{0} \mathrm{A}}{5}$
Download our appand get started for free
Similar Questions
- 1In a certain thermodynamical process, the pressure of a gas depends on its volume as $kV ^{3}$. The work done when the temperature changes from $100^{\circ} C$ to $300^{\circ} C$ will be .......... $nR$, where $n$ denotes number of moles of a gas.View Solution
- 2View SolutionWhich of the following can not determine the state of a thermodynamic system
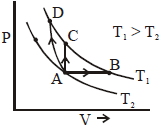
- 3Three different processes that can occur in an ideal monoatomic gas are shown in the $P$ vs $V$ diagram. The paths are labelled as $A \rightarrow B, A \rightarrow C$ and $A \rightarrow D .$ The change in internal energies during these process are taken as $E _{ AB }, E _{ AC }$ and $E _{ AD }$ and the work done as $W _{ AB }$ $W _{ AC }$ and $W _{ AD }$View Solution
The correct relation between these parameters are
- 4One mole of a monatomic ideal gas is taken through a cycle $ABCDA$ as shown in the $P-V$ diagram. Column $II$ gives the characteristics involved in the cycle. Match them with each of the processes qiven in Column $I$View Solution
Column $I$ Column $II$ $(A)$ Process $A \rightarrow B$ $(p)$ Internal energy decreases. $(B)$ Process $B \rightarrow C$ $(q)$ Internal energy increases. $(C)$ Process $C \rightarrow D$ $(r)$ Heat is lost. $(D)$ Process $D \rightarrow A$ $(s)$ Heat is gained. $(t)$ Work is done on the gas. 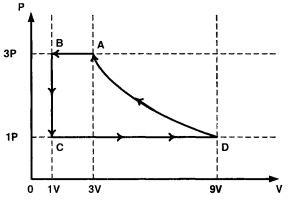
- 5View SolutionAn ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?
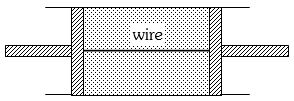
- 6A cylindrical tube of uniform cross-sectional area $A$ is fitted with two air tight frictionless pistons. The pistons are connected to each other by a metallic wire. Initially the pressure of the gas is $P_0$ and temperature is $T_0$, atmospheric pressure is also $P_0$. Now the temperature of the gas is increased to $2T_0$, the tension in the wire will beView Solution
- 7A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
- 8A heat engine is involved with exchange of heat of $1915\, J,$ $-40\, J ,+125\, J$ and $-Q\,J$, during one cycle achieving an efficiency of $50.0 \%$. The value of $Q$ is.......$J$View Solution
- 9An engine operating between the boiling and freezing points of water will haveView Solution
$1.$ efficiency more than $27 \%$
$2.$ efficiency less than the efficiency a Carnot engine operating between the same two temperatures.
$3.$ efficiency equal to $27 \%$
$4.$ efficiency less than $27 \%$
- 10A thermodynamic process in which temperature $T$ of the system remains constant though other variable $P$ and $V$ may change, is calledView Solution
