An engine operating between the boiling and freezing points of water will have
$1.$ efficiency more than $27 \%$
$2.$ efficiency less than the efficiency a Carnot engine operating between the same two temperatures.
$3.$ efficiency equal to $27 \%$
$4.$ efficiency less than $27 \%$
JEE MAIN 2023, Medium
$\eta=\left(1-\frac{273}{373}\right) \times 100=26.8 \%$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
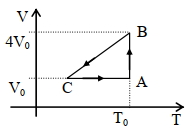
- 1One mole of an ideal gas in initial state $\mathrm{A}$ undergoes a cyclic process $A B C A$, as shown in the figure. Its pressure at $A$ is $\mathrm{P}_0$. Choose the correct option$(s)$ from the followingView Solution
$(A)$ Internal energies at $\mathrm{A}$ and $\mathrm{B}$ are the same
$(B)$ Work done by the gas in process $\mathrm{AB}$ is $\mathrm{P}_0 \mathrm{~V}_0 \ln 4$
$(C)$ Pressure at $C$ is $\frac{P_0}{4}$
$(D)$ Temperature at $\mathrm{C}$ is $\frac{\mathrm{T}_0}{4}$
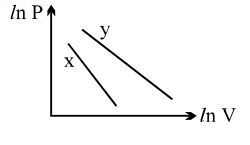
- 2For two different gases $X$ and $Y$, having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{V1}$ and $C_{V2}$ respectively, the ln $P$ versus ln $V$ graph is plotted for adiabatic process, as shownView Solution
- 3View SolutionOne mole of a monoatomic gas and one mole of a diatomic gas are initially in the same state. Both gases are expanded isothermally and then adiabatically, such that they acquire the same final state. Choose the correct statement.
- 4An ideal gas undergoes a polytropic given by equation $P V^n=$ constant. If molar heat capacity of gas during this process is arithmetic mean of its molar heat capacity at constant pressure and constant volume then value of $n$ is ..............View Solution
- 5For an adiabatic expansion of an ideal gas, the fractional change in its pressure is equal to (where $\gamma$ is the ratio of specific heats):View Solution
- 6A gas at $NTP$ is suddenly compressed to one-fourth of its original volume. If $\gamma $ is supposed to be $\frac{3}{2}$, then the final pressure is........ atmosphereView Solution
- 7View SolutionCan two isothermal curves cut each other
- 8View SolutionPressure versus temperature graph of an ideal gas is shown in figure
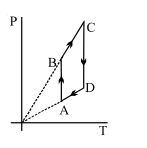
- 9View SolutionWhen heat in given to a gas in an isobaric process, then
- 10A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an absolute temperature $T$ and rejects heat to a sink at a temperature of $T/3$ . The amount of heat rejected isView Solution
