Given below are two statements : one is labelled as Assertion A and the other is labelled as Reason $R$. Assertion A : Efficiency of a reversible heat engine will be highest at $-273^{\circ} C$ temperature of cold reservoir.
Reason $R$ : The efficiency of Carnot's engine depends not only on temperature of cold reservoir but it depends on the temperature of hot reservoir too and is given as $\eta=\left(1-\frac{ T _2}{ T _1}\right)$.
In the light of the above statements, choose the correct answer from the options given below
JEE MAIN 2023, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If minimum possible work is done by a refrigerator in converting $100\; grams$ of water at $0^{\circ} C$ to ice, how much heat (in calories) is released to the surrounding at temperature $27^{\circ} C$ (Latent heat of ice $=80 Cal / gram$ ) to the nearest integer?View Solution
- 2View SolutionIn which of the following processes, heat is neither absorbed nor released by a system ?
- 3One mole of an ideal gas at an initial temperature of $T\, K$ does $6 R$ joules of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $5/3$, the final temperature of gas will beView Solution
- 4A Carnot engine, whose efficiency is $40\%$, takes in heat from a source maintained at a temperature of $500\ K$. It is desired to have an engine of efficiency $60\%$. Then, the intake temperature for the same exhaust (sink) temperature must be ....... $K$View Solution
- 5View SolutionFor which combination of working temperatures the efficiency of Carnot’s engine is highest
- 6View SolutionAn ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?
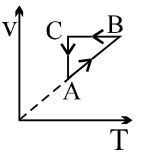
- 7During an isothermal expansion, a confined ideal gas does $-150 \,J$ of work against its surroundings. This implies thatView Solution
- 8View SolutionThe maximum possible efficiency of a heat engine is ...........
- 9One mole of an ideal gas having initial volume $V$, pressure $2P$ and temperature $T$ undergoes a cyclic process $ABCDA$ as shown below : The net work done in the complete cycle isView Solution
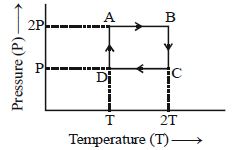
- 10A diatomic ideal gas is used in a carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\,V$ , the efficiency of the engine isView Solution
