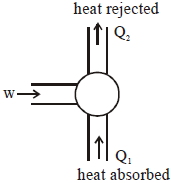
If minimum possible work is done by a refrigerator in converting $100\; grams$ of water at $0^{\circ} C$ to ice, how much heat (in calories) is released to the surrounding at temperature $27^{\circ} C$ (Latent heat of ice $=80 Cal / gram$ ) to the nearest integer?
JEE MAIN 2020, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will beView Solution
- 2$540$ calories of heat convert $1 $ cubic centimeter of water at ${100^o}C$ into $1671 $ cubic centimeter of steam at ${100^o}C$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly ...... $cal$View Solution
- 3An ideal gas heat engine operates in a Carnot cycle between $227^o C$ and $127^o C$. It absorbs $6\,kcal$ at the higher temperature. The amount of heat (in $kcal$) converted into work is equal toView Solution
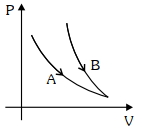
- 4Choose the correct statement for processes $A$ & $B$ shown in figure.View Solution
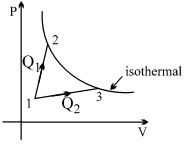
- 5A gas takes part in two processes in which it is heated from the same initial state $1$ to the same final temperature. The processes are shown on the $P-V$ diagram by the straight line $1-2$ and $1-3$. $2$ and $3$ are the points on the same isothermal curve. $Q_1$ and $Q_2$ are the heat transfer along the two processes. ThenView Solution
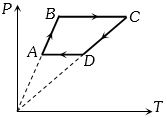
- 6Six moles of an ideal gas perfomrs a cycle shown in figure. If the temperature are $T_A = 600 K, T_B = 800 K, T_C = 2200 K \,and\, T_D = 1200 K$, the work done per cycle is ..... $kJ$View Solution
- 7A system is provided with $200 \,cal$ of heat and the work done by the system on the surrounding is $40 \,J$. Then its internal energyView Solution
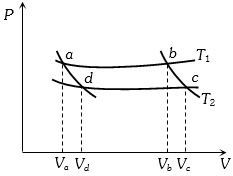
- 8In the following $P-V$ diagram two adiabatics cut two isothermals at temperatures $T_1$ and $T_2$ (fig.). The value of $\frac{{{V_a}}}{{{V_d}}}$ will beView Solution
- 9The efficiency of a Carnot engine operating with reservoir temperature of $100\,^{\circ} C$ and $-23\,^{\circ} C$ will beView Solution
- 10$0.02\, moles$ of an ideal diatomic gas with initial temperature $20^{\circ} C$ is compressed from $1500 \,cm ^{3}$ to $500 \,cm ^{3}$. The thermodynamic process is such that $p V^{2}=\beta$, where $\beta$ is a constant. Then, the value of $\beta$ is close to (the gas constant, $R=8.31 \,J / K / mol$ ).View Solution