Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Given that $1\,g$ of water in liquid phase has volume $1\,cm^3$ and in vapour phase $1671\, cm^3$ at atmospheric pressure and the latent heat of vaporization of water is $2256\,J/g;$ the change in the internal energy in joules for $1\,g$ of water at $373\,K$ when it changes from liquid phase to vapour phase at the same temperature is ....... $J$View Solution
- 2During an adiabatic expansion of $2\, moles$ of a gas, the change in internal energy was found $-50J.$ The work done during the process is ...... $J$View Solution
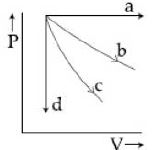
- 3View SolutionThe given diagram shows four processes i.e., isochoric, isobaric, isothermal and adiabatic. The correct assignment of the processes, in the same order is given by
- 4A piece of hot copper at $100^{\circ} C$ is plunged into a pond at $30^{\circ} C$. The copper cools down to $30^{\circ} C$, while the pond being huge stays at its initial temperature. Then,View Solution
- 5The specific heat at constant pressure of a real gas obeying $\mathrm{PV}^2=\mathrm{RT}$ equation is :View Solution
- 6$0.02\, moles$ of an ideal diatomic gas with initial temperature $20^{\circ} C$ is compressed from $1500 \,cm ^{3}$ to $500 \,cm ^{3}$. The thermodynamic process is such that $p V^{2}=\beta$, where $\beta$ is a constant. Then, the value of $\beta$ is close to (the gas constant, $R=8.31 \,J / K / mol$ ).View Solution
- 7An ideal gas expands in such a way that $PV^2 =$ constant throughout the process.View Solution
- 8View SolutionNeon gas of a given mass expands isothermally to double volume. What should be the further fractional decrease in pressure, so that the gas when adiabatically compressed from that state, reaches the original state?
- 9View SolutionFirst law thermodynamics states that
- 10$Assertion :$ In isothermal process whole of the heat supplied to the body is converted into internal energy.View Solution
$Reason :$ According to the first law of thermodynamics : $\Delta Q = \Delta U + p\Delta V$
