The efficiency of Carnot's engine operating between reservoirs, maintained at temperatures ${27^o}C$ and $ - {123^o}C,$ is ...... $\%$
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal gas expands adiabatically from an initial state $\left(T_A, V_0\right)$ to final state $\left(T_f, 5 V_0\right)$. Another mole of the same gas expands isothermally from a different initial state ( $T_{\mathrm{B}}, \mathrm{V}_0$ ) to the same final state $\left(T_{\mathrm{f}}, 5 V_0\right)$. The ratio of the specific heats at constant pressure and constant volume of this ideal gas is $\gamma$. What is the ratio $T_{\mathrm{A}} / T_{\mathrm{B}}$ ?View Solution
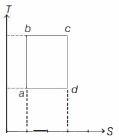
- 2An ideal gas is taken reversibly around the cycle $a-b-c-d-a$ as shown on the temperature $T$ - entropy $S$ diagram. The most appropriate representation of above cycle on a internal energy $U$ - volume $V$ diagram isView Solution
- 3View SolutionCan two isothermal curves cut each other
- 4In a thermodynamics process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20\,J$ of heat and $8\,J$ of work is done on the gas. If the initial internal energy of the gas was $30\,J$. The final internal energy will be...... $J$View Solution
- 5Consider the following volume-temperature $( V - T )$ diagram for the expansion of $5$ moles of an ideal monoatomic gas.View Solution
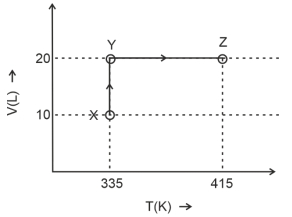
Considering only $P-V$ work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence $X \rightarrow Y \rightarrow Z$ is $\qquad$
[Use the given data: Molar heat capacity of the gas for the given temperature range, $C _{ v , m }=12 J K ^{-1} mol ^{-1}$ and gas constant, $R =8.3 J K ^{-1} mol ^{-1}$ ]
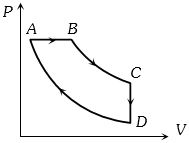
- 6A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same processView Solution
- 7$Assertion :$ The isothermal curves intersect each other at a certain point.View Solution
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope. - 8A Carnot engine whose sink is at $300 \,K$ has an efficiency of $50 \%$. By how much should the temperature of source be increased so as the efficiency becomes $70 \%$ is ............ $K$View Solution
- 9Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$View Solution
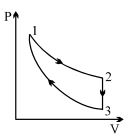
- 10An ideal heat engine exhausting heat at ${77^o}C$ is to have a $30\%$ efficiency. It must take heat at ...... $^oC$View Solution
