Two gases of equal mass are in thermal equilibrium. If ${P_a},\,{P_b}$ and ${V_a}$ and ${V_b}$ are their respective pressures and volumes, then which relation is true
AIIMS 1982, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionFor an isometric process
- 2View SolutionWhen heat is given to a gas in an isothermal change, the result will be
- 3For an adiabatic expansion of an ideal gas, the fractional change in its pressure is equal to (where $\gamma$ is the ratio of specific heats):View Solution
- 4A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
- 5A vessel containing $5\, litres$ of a gas at $0.8 \,pa$ pressure is connected to an evacuated vessel of volume $3$ litres. The resultant pressure inside will be ...... $pa$ (assuming whole system to be isolated)View Solution
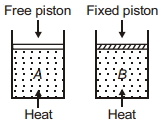
- 6Two cylinders contain same amount of ideal monatomic gas. Same amount of heat is given to two cylinders. If temperature rise in cylinder $A$ is $T_0$ then temperature rise in cylinder $B$ will be .........View Solution
- 7$110\; J$ of heat is added to a gaseous system, whose internal energy change is $40\; J$, then the amount of external work done is ........ $J$View Solution
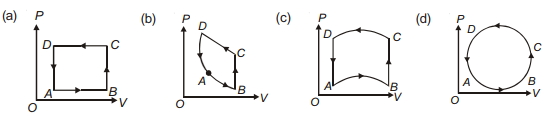
- 8In following figures $(a)$ to $(d)$, variation of volume by change of pressure is shown in figure. The gas is taken along the path $A B C D A$. Change in internal energy of the gas will be .......View Solution
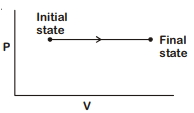
- 9The $P-V$ diagram for an ideal gas in a piston cylinder assembly undergoing a thermodynamic process is shown in the figure. The process isView Solution
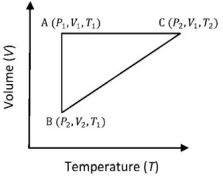
- 10A reversible cyclic process for an ideal gas is shown below. Here, $P, V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
(image)
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$
$(B)$ $\mathrm{W}_{\mathrm{BC}}=\mathrm{P}_2\left(\mathrm{~V}_2-\mathrm{V}_1\right)$ and $\mathrm{q}_{\mathrm{BC}}=\mathrm{H}_{\mathrm{AC}}$
$(C)$ $\Delta \mathrm{H}_{\mathrm{CA}}<\Delta \mathrm{U}_{\mathrm{CA}}$ and $\mathrm{q}_{\mathrm{AC}}=\Delta \mathrm{U}_{\mathrm{BC}}$
$(D)$ $\mathrm{q}_{\mathrm{BC}}=\Delta \mathrm{H}_{\mathrm{AC}}$ and $\Delta \mathrm{H}_{\mathrm{CA}}>\Delta \mathrm{U}_{\mathrm{CA}}$