$T _1=127^{\circ}\,C =400\,K$
$T _2=527^{\circ}\,C =800\,K$
Efficiency $=\eta=1-\frac{T_1}{T_2}$
$=1-\frac{400}{800}=\frac{1}{2}$
Percentage efficiency $=\frac{1}{2} \times 100=50 \%$
Download our appand get started for free
Similar Questions
- 1A gas ($\gamma = 1.3)$ is enclosed in an insulated vessel fitted with insulating piston at a pressure of ${10^5}\,N/{m^2}$. On suddenly pressing the piston the volume is reduced to half the initial volume. The final pressure of the gas isView Solution
- 2View SolutionThe first law of thermodynamics is concerned with the conservation of
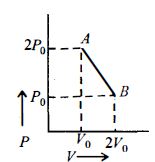
- 3Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternative.View Solution
- 4$Assertion :$ Thermodynamic process in nature are irreversible.View Solution
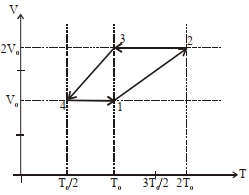
$Reason :$ Dissipative effects cannot be eliminated. - 5One mole of a monoatomic ideal gas goes through a thermodynamic cycle, as shown in the volume versus temperature ($V-T$) diagram. The correct statement($s$) is/are :View Solution
[ $R$ is the gas constant]
$(1)$ Work done in this thermodynamic cycle $(1 \rightarrow 2 \rightarrow 3 \rightarrow 4 \rightarrow 1)$ is $| W |=\frac{1}{2} RT _0$
$(2)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $2 \rightarrow 3$ is $\left|\frac{ Q _{1 \rightarrow 2}}{ Q _{2 \rightarrow 3}}\right|=\frac{5}{3}$
$(3)$ The above thermodynamic cycle exhibits only isochoric and adiabatic processes.
$(4)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $3 \rightarrow 4$ is $\left|\frac{Q_{U \rightarrow 2}}{Q_{3 \rightarrow 4}}\right|=\frac{1}{2}$
- 6View SolutionIn a thermodynamic system working substance is ideal gas, its internal energy is in the form of
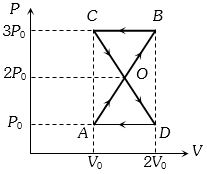
- 7A thermodynamic system undergoes cyclic process $ABCDA $ as shown in figure. The work done by the system in the cycle isView Solution
- 8View SolutionWhich is incorrect
- 9When an ideal gas $(\gamma = 5/3$) is heated under constant pressure, then what percentage of given heat energy will be utilised in doing external workView Solution
- 10$n\, moles$ of an ideal gas undergo a process $A \to B$ as shown in the figure. Maximum temperature of the gas during the process isView Solution