If a cylinder containing a gas at high pressure explodes, the gas undergoes
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
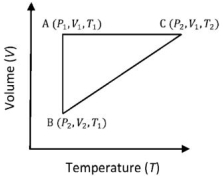
- 1A reversible cyclic process for an ideal gas is shown below. Here, $P , V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$ $(B)$ $W _{ BC }= P _2\left( V _2- V _1\right)$ and $q _{ BC }= H _{ AC }$ $(C)$ $\Delta H _{ CA }<\Delta U _{ CA }$ and $q _{ AC }=\Delta U _{ BC }$ $(D)$ $q_{B C}=\Delta H_{A C}$ and $\Delta H_{C A}>\Delta U_{C A}$
- 2During the adiabatic expansion of $2 \,moles$ of a gas, the internal energy was found to have decreased by $100 J$. The work done by the gas in this process is ..... $J$View Solution
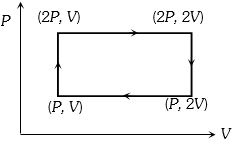
- 3Work done in the given $P-V$ diagram in the cyclic process isView Solution
- 4A given ideal gas with $\gamma = \frac{{{C_p}}}{{{C_v}}} = 1.5$ at a temperature $T$. If the gas is compressed adiabatically to one-fourth of its initial volume, the final temperature will be ..... $T$View Solution
- 5A cylinder fitted with a piston contains $0.2 \,moles$ of air at temperature $27°C.$ The piston is pushed so slowly that the air within the cylinder remains in thermal equilibrium with the surroundings. Find the approximate work done by the system if the final volume is twice the initial volume ...... $J$View Solution
- 6A sample of an ideal gas is contained in a cylinder. The volume of the gas is suddenly decreased. A student makes the following statements to explain the change in pressure of the gasView Solution
$I.$ The average kinetic energy of the gas atoms increases
$II.$ The atoms of the gas hit the walls of the cylinder more frequently
$III.$ Temperature of the gas remains unchanged
Which of these statements is true? - 7A reversible engine has an efficiency of $\frac{1}{4}$. If the temperature of the sink is reduced by $58^{\circ} {C}$, its efficiency becomes double. Calculate the temperature of the sink. (In $^{\circ} {C}$)View Solution
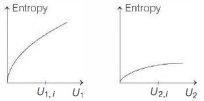
- 8Graphs below show the entropy versus energy $U$ of two systems $1$ and $2$ at constant volume. The initial energies of the systems are indicated by $U_{1, i}$ and $U_{2, i}$, respectively. Graphs are drawn to the same scale. The systems are then brought into thermal contact with each other. Assume that, at all times the combined energy of the two systems remains constant. Choose the most appropriate option indicating the energies of the two systems and the total entropy after they achieve the equilibrium.View Solution
- 9View SolutionThe specific heat of a gas at constant pressure is more than that of the same gas at constant volume because
- 10View SolutionAn ideal diatomic gas is heated at constant pressure. The ratio of the work done to the heat supplied is
