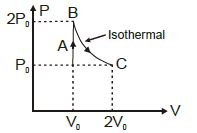
Work done in the given $P-V$ diagram in the cyclic process is
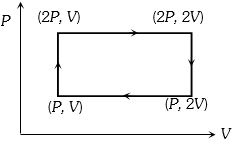

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A scientist says that the efficiency of his heat engine which operates at source temperature $127°C$ and sink temperature $27°C$ is $26\%$, thenView Solution
- 2View SolutionIn thermodynamic processes which of the following statements is not true?
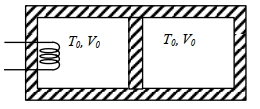
- 3A thermally insulating cylinder has a thermally insulating and frictionless movable partition in the middle, as shown in the figure below. On each side of the partition, there is one mole of an ideal gas, with specific heat at constant volume, $C_v=2 R$. Here, $R$ is the gas constant. Initially, each side has a volume $V_0$ and temperature $T_0$. The left side has an electric heater, which is turned on at very low power to transfer heat $Q$ to the gas on the left side. As a result the partition moves slowly towards the right reducing the right side volume to $V_0 / 2$. Consequently, the gas temperatures on the left and the right sides become $T_L$ and $T_R$, respectively. Ignore the changes in the temperatures of the cylinder, heater and the partition.View Solution
($1$) The value of $\frac{T_R}{T_0}$ is
$(A)$ $\sqrt{2}$ $(B)$ $\sqrt{3}$ $(C)$ $2$ $(D)$ $3$
($2$) The value of $\frac{Q}{R T_0}$ is
$(A)$ $4(2 \sqrt{2}+1)$ $(B)$ $4(2 \sqrt{2}-1)$ $(C)$ $(5 \sqrt{2}+1)$ $(D)$ $(5 \sqrt{2}-1)$
Give the answer or qution ($1$) and ($2$)
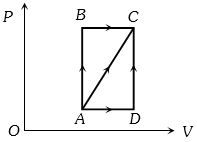
- 4A thermodynamic process is shown in the figure. The pressures and volumes corresponding to some points in the figure are :View Solution
${P_A} = 3 \times {10^4}Pa,\;{P_B} = 8 \times {10^4}Pa$ and ${V_A} = 2 \times {10^{ - 3}}{m^3},\;{V_D} = 5 \times {10^{ - 3}}{m^3}$
In process $AB$, $600 J$ of heat is added to the system and in process $BC, 200 J $ of heat is added to the system. The change in internal energy of the system in process $ AC$ would be ...... $J$
- 5A Carnot engine absorbs $1000\,J$ of heat energy from a reservoir at $127\,^oC$ and rejects $600\,J$ of heat energy during each cycle. The efficiency of engine and temperature of sink will beView Solution
- 6The heat capacity of one mole an ideal is found to be $C_V=3 R(1+a R T) / 2$, where $a$ is a constant. The equation obeyed by this gas during a reversible adiabatic expansion isView Solution
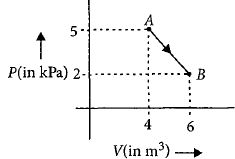
- 7One mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure.View Solution
The change in internal energy of the gas during the transition is ............$\;kJ$
- 8A polyatomic gas with six degrees of freedom does $25\,\,J$ of work when it is expanded at constant pressure. The heat given to the gas is ..... $J$View Solution
- 9A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an abosolute temperature $T$ and rejects heat to a sink at a temperature of $T/3.$ The amount of heat rejected isView Solution
- 10A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$View Solution